
1,4-Butanediol diglycidyl ether synthesis
- Product Name:1,4-Butanediol diglycidyl ether
- CAS Number:2425-79-8
- Molecular formula:C10H18O4
- Molecular Weight:202.25
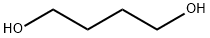
110-63-4
703 suppliers
$24.80/250g

106-89-8
752 suppliers
$9.00/10g

2425-79-8
352 suppliers
$30.00/5g
Yield:2425-79-8 89%
Reaction Conditions:
with tetrabutyl-ammonium chloride;water;sodium hydroxide at 40; for 0.75 h;
Steps:
2.3. General method for synthesis of diglycidyl ether intermediates
General procedure: Diglycidyl ether intermediates were prepared under phase-transfercatalytic condition: To the vigorously stirred mixture of epichlorohydrin(9.34 mL, 120 mmol), sodium hydroxide pellets (4.8 g,120 mmol), water (0.5 mL, 28 mmol), and tetrabutylammoniumchloride (0.28 g, 1 mmol), cooled Glycol (20 mmol, 1,2-ethanediol or1,4-butanediol) was added dropwise. After the completion of the addition,the mixture was stirred for 45 min at 40 °C. The solid productsthat were formed alongside the reaction removed by filtration andwashed with CH2Cl2 (30 mL×3). The combined organic layers weredried over anhydrous Na2SO4 and additional dichloromethane andexcess epichlorohydrin evaporated off by distillation to give a yellowoil. The resulting oil was purified by distillation under reduced pressureto give a pure and colorless product (89% yields).
References:
Abbasi, Hassan;Pourrostam-Ravadanaq, Pariya;Safa, Kazem D. [Analytical Biochemistry,2020,vol. 597]