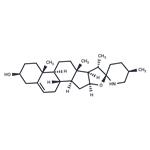
- Solasodine
-
- $68.00 / 20mg
-
2024-11-19
- CAS:126-17-0
- Min. Order:
- Purity: 99.94%
- Supply Ability: 10g
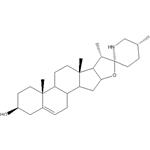
- SOLASODINE
-
- $0.00 / 1kg
-
2024-11-06
- CAS:126-17-0
- Min. Order: 0.10000000149011612kg
- Purity: ≥98%HPLC
- Supply Ability: 20tons
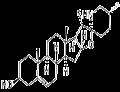
- Solasodine
-
- $0.00 / 20mg
-
2023-02-24
- CAS:126-17-0
- Min. Order: 5mg
- Purity: ≥98%(HPLC)
- Supply Ability: 10 g
|
| | SOLASODINE Basic information |
| | SOLASODINE Chemical Properties |
| Melting point | 200-202° | | alpha | D25 -98° (c = 0.14 in methanol); D -113° (CHCl3) | | Boiling point | 532.75°C (rough estimate) | | density | 1.0159 (rough estimate) | | refractive index | 1.6400 (estimate) | | storage temp. | 2-8°C | | solubility | DMSO: soluble0.5mg/mL, clear (warmed) | | pka | pKb 6.30(at 25℃) | | form | powder | | color | white to beige | | optical activity | [α]/D -88 to -98°, c = 0.2 in methanol | | InChIKey | KOZCINYDCJVLDW-GCGBSLFCSA-N | | CAS DataBase Reference | 126-17-0(CAS DataBase Reference) | | NIST Chemistry Reference | Solasodine(126-17-0) |
| | SOLASODINE Usage And Synthesis |
| Description | There is still some doubt as to whether this base occurs as such in Solanum
aviculare and S. xanthocarpum or whether it is an artifact formed from Sola_x0002_sonine (q.v.) during the extraction process. The base is laevorotatory having [α]20D - 92.4° (C6H6) and the following salts and derivatives have been prepared:
hydrochloride, m.p. 314°C; hydriodide, m.p. 293°C; oxalate, m.p. 248°C; tart�rate, m.p. 222°C; picrate, m.p. 1.4l-2°C; picrolonate, m.p. 234°C; acetyl
derivative, m.p. 195°C; benzoyl compound, m.p. 2l6-7°C and the 3:5-dinitro�benzoyl derivative, m.p. 191.5-193°C. The alkaloid forms a sparingly soluble
digitonide and yields Diels's hydrocarbon on selenium dehydrogenation. | | Uses | A potentially useful precursor. | | Uses | antineoplastic, antiinflammatory | | Uses | Solasodine is a potentially useful precursor for steroidal compounds and hormones, this is usually found in plants from the solanaceae family. It also has potent cytotoxic and anti-inflammatory effects. | | Uses | Starting material for steroidal drugs. | | Definition | ChEBI: Solasodine is an oxaspiro compound and steroid alkaloid sapogenin with formula C27H43NO2 found in the Solanum (nightshade) family. It is used as a precursor in the synthesis of complex steroidal compounds such as contraceptive pills. It has a role as a plant metabolite, a teratogenic agent, a diuretic, an antifungal agent, a cardiotonic drug, an immunomodulator, an antipyretic, an apoptosis inducer, an antioxidant, an antiinfective agent, an anticonvulsant, a central nervous system depressant and an antispermatogenic agent. It is an azaspiro compound, an oxaspiro compound, an alkaloid antibiotic, a hemiaminal ether, a sapogenin and a steroid alkaloid. It is a conjugate base of a solasodine(1+). | | General Description | This substance is a primary reference substance with assigned absolute purity (considering chromatographic purity, water, residual solvents, inorganic impurities). The exact value can be found on the certificate. Produced by PhytoLab GmbH & Co. KG | | Biochem/physiol Actions | Solasodine is a neuroprotective antioxidant glycoalkaloid of Solanum species. Solasodine increases superoxide dismutase (SOD), catalase (CAT),and glutathione (GSH) levels, while reducing lipid peroxidation (LPO) and nitric oxide (NO) levels in the brains of ischemia/reperfusion (I/R)-injury model in rats. Also, Solasonine displayed multiple activities including, anti-cancer and leishmanicidal activities. | | Safety Profile | Poison by intraperitoneal route. Moderately toxic by ingestion. Experimental teratogenic and reproductive effects. When heated to decomposition it emits toxic fumes of NOx. | | Purification Methods | Solasodine crystallises (as monohydrate) from MeOH as lustrous plates (on heating, the plates change to needles as they melt and resolidify in needles), or aqueous 80% EtOH, and sublimes at high vacuum. After recrystallisation from H2O, m 198-199o, then from Me2CO/H2O, m 199-201o, it has [] D 22 -109.3o (c 0.581, CHCl3). On TLC with silica gel G (*C6H6/absolute EtOH, 8:2) it has RF 0.45. IR (KBr): max at 10.3, 10.4, 11.2, 11.5 (azaoxaspirane bands). [Schreiber & Rnsch Tetrahedron 20 1939 1964, Uhle J Org Chem 27 656 1962, Kessar et al. Tetrahedron 27 2153 1971, Beilstein 27 III/IV 2000.] | | References | Rochelmeyer, Chen., Arch. Pharm., 277,329 (1939)
Briggs., J. Chem. Soc., 3, (1942)
Briggs et al., ibid, 3013 (1950)
Absolute configuration:
Boll, Phillipsborn., Acta Chem. Scand., 19, 1365 (1965)
Synthesis:
Schreiber, Walther, Ronsch., Tetrahedron, 20, 1939 (1964)
Adam, Schreiber., Experientia, 21,471 (1965)
Adam, Schreiber., Tetrahedron, 22,3591 (1966) |
| | SOLASODINE Preparation Products And Raw materials |
| Raw materials | Cryptogenin-->Pregn-5-ene-3,16-diol, 20-[(5R)-3,4,5,6-tetrahydro-5-methyl-2-pyridinyl]-, 3,16-diacetate, (3β,16β,20S)--->Solasodine, hydrochloride-->Furosta-5,20(22)-dien-3-ol, 26-amino-, (3β,25R)--->(20R,25R)-spirost-5-en-3beta-yl acetate | | Preparation Products | (22R,25R)-Spirosol-4-en-3-one |
|