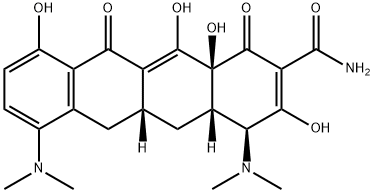
Minocycline
|
|
Minocycline Eigenschaften
- alpha
- D25 -166° (c = 0.524)
- Siedepunkt:
- 563.31°C (rough estimate)
- Dichte
- 1.3283 (rough estimate)
- Brechungsindex
- 1.6500 (estimate)
- L?slichkeit
- Methanol (Slightly)
- Aggregatzustand
- Solid
- pka
- pKa 2.8 (Uncertain);5.0 (Uncertain);7.8 (Uncertain);9.5 (Uncertain)
- Farbe
- Yellow to Dark Orange
- Wasserl?slichkeit
- 52g/L(25 ºC)
- Stabilit?t:
- Light Sensitive
- CAS Datenbank
- 10118-90-8(CAS DataBase Reference)
- EPA chemische Informationen
- Minocycline (10118-90-8)
Sicherheit
- Risiko- und Sicherheitserkl?rung
- Gefahreninformationscode (GHS)
| Giftige Stoffe Daten | 10118-90-8(Hazardous Substances Data) |
|---|
| Bildanzeige (GHS) |
 
|
||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alarmwort | Warnung | ||||||||||||||||||||||||||||
| Gefahrenhinweise |
|
||||||||||||||||||||||||||||
| Sicherheit |
|
Minocycline Chemische Eigenschaften,Einsatz,Produktion Methoden
Beschreibung
An important antibiotic produced by semisynthesis from demeclocycline is minocycline. It is much more lipophilic than its precursors, gives excellent blood levels following oral administration (90–100% available),and can be given once a day. Its absorption is lowered by approximately 20% when taken with food or milk. It is less dependent on active uptake mechanisms and has a somewhat broader antimicrobial spectrum. It also, apparently, is less painful on IM or IV injection, but it has vestibular toxicities (e.g., vertigo, ataxia, and nausea) not generally shared by other tetracyclines.Verwenden
Minocycline is a semi-synthetic tetracycline prepared by sequential hydrogenolysis, nitration and reductive methylation. Minocycline, together with doxycycline, is regarded as a ‘third generation’ tetracycline largely replacing the natural products and pro-drugs produced in the early 1950s for mainstream antibiotic applications. Like all tetracyclines, minocycline shows broad spectrum antibacterial and antiprotozoan activity and acts by binding to the 30S and 50S ribosomal sub-units, blocking protein synthesis. Minocycline has been extensively cited in the literature with over 5,000 references.Definition
ChEBI: A tetracycline analogue having a dimethylamino group at position 7 and lacking the methyl and hydroxy groups at position 5.Indications
The tetracycline antibiotic minocycline (Minocin) is modestly effective in the treatment of rheumatoid arthritis and is generally well tolerated. Radiographic evidence of its efficacy as a DMARD is lacking, although clinical symptoms do abate. It can be useful in the treatment of early, mild disease.Weltgesundheitsorganisation (WHO)
Minocycline, a semi-synthetic tetracycline derivative was introduced in 1967. It is used today in the treatment of bacterial, rickettsial and amoebic infections. Symptoms described as dizziness or vertigo have been recognized in association with minocycline administration, however, these symptoms are usually not severe. Minocycline is registered in many countries and the World Health Organization is not aware that registration has been refused elsewhere.Antimicrobial activity
It exhibits the broad-spectrum activity typical of the group, but retains activity against some strains of Staph. aureus resistant to older tetracyclines. It is active against β-hemolytic streptococci and some tetracycline- resistant pneumococci. It is also active against some enterobacteria resistant to other tetracyclines, probably because some Gram-negative efflux pumps remove minocycline less effectively than other tetracyclines. Some strains of H. influenzae resistant to other tetracyclines are susceptible. Sten. maltophilia is susceptible, as are most strains of Acinetobacter spp. and L. pneumophila.It is notable for its activity against Bacteroides and Fusobacterium spp., and is more active than other tetracyclines against C. trachomatis, brucellae and nocardiae. It inhibits Mycobacterium tuberculosis, M. bovis, M. kansasii and M. intracellulare at 5–6 mg/L. Candida albicans and C. tropicalis are also slightly susceptible.
Pharmazeutische Anwendungen
A semisynthetic tetracycline derivative supplied as the hydrochloride for oral administration.Pharmakokinetik
Oral absorption: 95–100%Cmax 150 mg oral: 4 mg/L after 2h
300 mg oral: 2 mg/L after 2 h
Plasma half-life: 12–24 h
Volume of distribution: 80–115 L
Plasma protein binding: 76%
Absorption
Food does not significantly affect absorption, which is depressed by co-administration with milk. It is chelated by metals and suffers the effects of antacids and ferrous sulfate common to tetracyclines. On a regimen of 100 mg every 12 h, steady-state concentrations ranged between 2.3 and 3.5 mg/L.
Distribution
The high lipophilicity of minocycline provides wide distribution and tissue concentrations that often exceed those of the plasma. The tissue:plasma ratio in maxillary sinus and tonsillar tissue is 1.6: that in lung is 3–4. Sputum concentrations may reach 37–60% of simultaneous plasma levels. In bile, liver and gallbladder the ratios are 38, 12 and 6.5, respectively.
Prostatic and seminal fluid concentrations range from 40% to 100% of those of serum. CSF penetration is poor, especially in the non-inflamed state. Concentrations in tears and saliva are high, and may explain its beneficial effect in the treatment of meningococcal carriage.
Metabolism
Biotransformation to three microbiologically inactive metabolites occurs in the liver: the most abundant is 9-hydroxyminocycline.
Excretion
Only 4–9% of administered drug is excreted in the urine, and in renal failure elimination is little affected. Neither hemodialysis nor peritoneal dialysis affects drug elimination. Fecal excretion is relatively low and evidence for enterohepatic recirculation remains uncertain. Despite high hepatic excretion, dose accumulation does not occur in liver disease, such as cirrhosis. Type IIa and type IV hyperlipidemic patients show a decreased minocycline clearance of 50%, suggesting that dose modification may be necessary.
Clinical Use
There appear to be few situations in which it has a unique therapeutic advantage over other tetracyclines. Its use has been tempered by the high incidence of vestibular side effects.Although used in the long-term management of acne, the potential for skin pigmentation must be considered. Because of its high tissue concentrations, it may occasionally provide a useful alternative to other agents for the treatment of chronic prostatitis. It has a role in the treatment of sexually transmitted chlamydial infections.
Nebenwirkungen
Minocycline shares the untoward reactions common to the group with gastrointestinal side effects being most common, and more prevalent in women. Diarrhea is relatively uncommon, presumably as a result of its lower fecal concentrations. Hypersensitivity reactions, including rashes, interstitial nephritis and pulmonary eosinophilia, are occasionally seen.Staining of the permanent dentition occurs with all tetracyclines; a side effect that appears to be unique to minocycline is that of tissue discoloration and skin pigmentation. Tissues that may become pigmented include the skin, skull and other bones and the thyroid gland, which at autopsy appears blackened. The pigmentation tends to resolve slowly with discontinuation of the drug and is related to the length of therapy.
Three types of pigmentation have been identified:
? A brown macular discoloration (‘muddy skin syndrome’), which occurs in sun-exposed parts and is histologically associated with melanin deposition.
? Blue–black macular pigmentation occurring within inflamed areas and scars associated with hemosiderin deposition.
? Circumscribed macular blue–gray pigmented areas occurring in sun-exposed and unexposed skin, which appears to be linked to a breakdown product of minocycline.
CNS toxicity has been prominent, notably benign intracranial hypertension, which resolves on discontinuation of the drug, and, more commonly, dizziness, ataxia, vertigo, tinnitus, nausea and vomiting, which appear to be more frequent in women. These primarily vestibular side effects have ranged in frequency from 4.5% to 86%. They partly coincide with plasma concentration peaks, but their exact pathogenesis has yet to be determined.
Minocycline Upstream-Materialien And Downstream Produkte
Upstream-Materialien
Wasserstoff
Demecycline
Dibenzylazodicarboxylat
7-Iodo Sancycline
Tigecycline Metabolite M6 (9-AniMoMinocycline)
(Dimethylamino)trimethylstannan
Demeclocyclinhydrochlorid
Azetidin
Downstream Produkte
Minocycline Anbieter Lieferant Produzent Hersteller Vertrieb H?ndler.
Global( 179)Lieferanten
| Firmenname | Telefon | Land | Produktkatalog | Edge Rate | |
|---|---|---|---|---|---|
| Hebei Mojin Biotechnology Co., Ltd | +86 13288715578 +8613288715578 |
sales@hbmojin.com | China | 12832 | 58 |
| Capot Chemical Co.,Ltd. | +86-(0)57185586718 +86-13336195806 |
sales@capot.com | China | 29793 | 60 |
| Henan Tianfu Chemical Co.,Ltd. | +86-0371-55170693 +86-19937530512 |
info@tianfuchem.com | China | 21634 | 55 |
| ATK CHEMICAL COMPANY LIMITED | +undefined-21-51877795 |
ivan@atkchemical.com | China | 32956 | 60 |
| career henan chemical co | +86-0371-86658258 +8613203830695 |
sales@coreychem.com | China | 29880 | 58 |
| Zhejiang ZETian Fine Chemicals Co. LTD | +8618957127338 |
stella@zetchem.com | China | 2136 | 58 |
| Hebei Weibang Biotechnology Co., Ltd | +8615531157085 |
abby@weibangbio.com | China | 8806 | 58 |
| Chongqing Chemdad Co., Ltd | +86-023-6139-8061 +86-86-13650506873 |
sales@chemdad.com | China | 39894 | 58 |
| CONIER CHEM AND PHARMA LIMITED | +8618523575427 |
sales@conier.com | China | 49374 | 58 |
| Shaanxi Dideu Medichem Co. Ltd | +86-29-87569266 15319487004 |
1015@dideu.com | China | 3940 | 58 |
10118-90-8()Verwandte Suche:
4-Dimethylamino-benzaldehyd
Dimethylammoniumchlorid
Benzyldimethylamin
Tetracyclin
Glycin
N,N-Dimethyl-1,3-diaminopropan
N-Methylmethanamin, gasf?rmig
1,3-Benzoldimethanamin
Aminocapronsure
ALTRENOGEST
3,6-Bis[(4-chlor-2-phosphonophenyl)azo]-4,5-dihydroxynaphthalin-2,7-disulfonsure
Trometamol
N,N-Dimethylpyridin-4-amin
Dimethylhydrogenphosphat
2-Dimethylaminoethanol
[4S-(4α,4aα,5aα,12aα)]-4,7-Bis(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a-tetrahydroxy-1,11-dioxonaphthacen-2-carboxamidmonohydrochlorid
TRIS EDTA ACETATE BUFFER, 10X, DNASE, RNASE AND PROTEASE FREE, PH 8.3, FOR MOLECULAR BIOLOGY
Naphthacen
- 7-DIMETHYLAMINO-6-DEMETHYL-6-DEOXYTETRACYCLINE HYDROCHLORIDE
- 7-DIMETHYLAMINO-6-DEMETHYL-6-DEOXYTETRACYCLINE, HCL
- MYNOCINE
- MINOCIN
- MINOCYCLINE
- MinocyclineBase
- Minocycline Hcl 13614-98-7 / Base
- 2-Naphthacenecarboxamide, 4,7-bis(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a-tetrahydroxy-1,11-dioxo- (8CI)
- 2-Naphthacenecarboxamide, 4,7-bis(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a-tetrahydroxy-1,11-dioxo-, (4S,4aS,5aR,12aS)- (9CI)
- 2-Naphthacenecarboxamide, 4,7-bis(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a-tetrahydroxy-1,11-dioxo-, [4S-(4a,4aa,5aa,12aa)]-
- (4S,4aS,5aR,12aS)-4,7-Bis(dimethylamino)-3,10,12,12a-tetrahydroxy-1,11-dioxo-1,4,4a,5,5a,6,11,12a-octahydrotetracene-2-carboxamide
- (4S)-4β,7-Bis(dimethylamino)-1,4,4aβ,5,5aβ,6,11,12a-octahydro-3,10,12,12aβ-tetrahydroxy-1,11-dioxo-2-naphthacenecarboxamide
- 4β,7-Bis(dimethylamino)-1,4,4aβ,5,5aβ,6,11,12a-octahydro-3,10,12,12aβ-tetrahydroxy-1,11-dioxo-2-naphthacenecarboxamide
- Minocycline hydrochlorid
- 7-DiMethylaMino-6-deMethyl-6-deoxytetracycline, CL 59806, Minocyclin
- Minocycline dihydrochloride
- 12,12a-tetrahydroxy-1,11-dioxo--10
- 2-naphthacenecarboxamide,4,7-bis(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro
- -3,10,12,12a-tetrahydroxy-1,11,-dioxo-,(4s-(4alpha,4aalpha,5aalpha,12aalpha))
- 7-dimethylamino-6-demethyl-6-deoxytetracycline
- cl59806
- (2e,4s,4ar,5as,12ar)-2-(amino-hydroxy-methylidene)-4,7-bis(dimethylamino)-10,11,12a-trihydroxy-4a,5,5a,6-tetrahydro-4h-tetracene-1,3,12-trione
- CS-588
- Tigecycline Impurity 32
- 2-Naphthacenecarboxamide, 4,7-bis(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a-tetrahydroxy-1,11-dioxo-, (4S,4aS,5aR,12aS)-
- (4S,4As,5ar,12as)-4,7-bis(dimethylamino)-3,10,12,12a-tetrahydroxy-1,11-dioxo-4a,5,5a,6-tetrahydro-4h-tetracene-2-carboxamide dihydrochloride
- 9-Monodemethyl Minocycline (Crude)
- Tigecycline Impurity (Minocycline)
- Tigecycline EP impurity CQ: What is Tigecycline EP impurity C Q: What is the CAS Number of Tigecycline EP impurity C
- MINOCYCLINE HCL
- [4s-(4alpha,4aalpha,5aalpha,12aalpha)]-4,7-bis(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a-tetrahydroxy-1,11-dioxonaphthacene-2-carboxamide monohydrochloride
- minocyclin
- Minocycline USP/EP/BP
- Tigecycline EP Impurity C(Minocycline)
- Omadacycline minocycline
- Tigecycline Impurity 22 (Tigecycline EP Impurity C ) (Minocycline)
- Adrenaline Impurity 55
- 10118-90-8
- C23H27N3O72ClH
- Antibiotics
- Antibiotics G-M
- Antibiotics A to Z
- BioChemical