| Identification | More | [Name]
Lactose | [CAS]
14641-93-1 | [Synonyms]
4-O-BETA-D-GALACTOPYRANOSYL-ALPHA-D-GLUCOSE
4-O-BETA-D-GALACTOPYRANOSYL-ALPHA-D-GLUCOSE MONOHYDRATE
A-4-(B-D-GALACTOSIDO)-D-GLUCOSE
A-LACTIN
A-LACTOBIOSE
ALPHA-D(+)-LACTOBIOSE MONOHYDRATE
ALPHA-D-LACTOSE
ALPHA-D-(+)-LACTOSE, HYDRATE
ALPHA-D(+)-LACTOSE MONOHYDRATE
ALPHA-D-LACTOSE MONOHYDRATE
ALPHA-LACTOSE MONOHYDRATE
A-MILK SUGAR
A-SACCHARUM LACTIN
BETA-D-GAL-(1->4)-ALPHA-D-GLC
BETA-D-GAL-(1->4)-ALPHA-D-GLC, MONOHYDRATE
D(+)-LACTOSE HYDRATE
D-(+)-LACTOSE HYDRATE,ALPHA-FORM
D-(+)-LACTOSE, MONOHYDRATE
D-LACTOSE MONOHYDRATE
LACTOSE MONOHYDRATE | [EINECS(EC#)]
238-691-8 | [Molecular Formula]
C12H22O11 | [MDL Number]
MFCD00150747 | [Molecular Weight]
342.3 | [MOL File]
14641-93-1.mol |
| Chemical Properties | Back Directory | [Melting point ]
238 °C | [Boiling point ]
667.9±55.0 °C(Predicted) | [density ]
1.76±0.1 g/cm3(Predicted) | [refractive index ]
52.5 ° (C=10, H2O) | [storage temp. ]
Inert atmosphere,Room Temperature | [form ]
Solid | [pka]
12.02±0.70(Predicted) | [color ]
White to light yellow | [Merck ]
5343 | [InChI]
InChI=1/C12H22O11/c13-1-3-5(15)6(16)9(19)12(22-3)23-10-4(2-14)21-11(20)8(18)7(10)17/h3-20H,1-2H2/t3-,4-,5-,6+,7-,8-,9-,10-,11?,12-/s3 | [InChIKey]
GUBGYTABKSRVRQ-JGYSHAQPNA-N | [SMILES]
[C@@H]1([C@H](O)[C@H](C(O)O[C@@H]1CO)O)O[C@@H]1[C@H](O)[C@H]([C@H](O)[C@@H](CO)O1)O |&1:0,1,3,7,12,13,15,16,18,r| | [CAS DataBase Reference]
14641-93-1(CAS DataBase Reference) | [EPA Substance Registry System]
14641-93-1(EPA Substance) |
| Questions and Answers (Q&A) | Back Directory | [General Description]
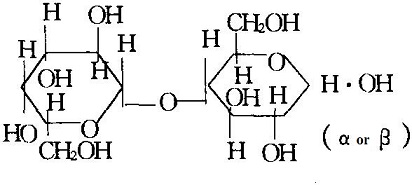
In nature, it only exists in mammalian milk. It belongs to disaccharides, being composed of one molecule of glucose and one molecule of galactose with a sweetness of about 16. Milk contains about 4.3% ~ 4.5% of lactose. There are two isomers of lactose, namely, α-and β-type, being monohydrate and anhydrous, respectively. The α-lactose usually contains one molecule of crystal water with the melting point of 202 °C; β-type is as an anhydride with the melting point of 252 °C and has a higher sweetness and higher solubility than α-type. Lactose is made from whey, a by-product of cheese or casein, and is used in large quantities as a drug excipient, as a nutrient for infants and young children, and as a good medium for penicillin.
It is used as a diluent for soluble powder dispersions, oral capsules, powder inhalers and tablets; lactose is also used in lyophilisates and intravenous injections.
|
| Questions And Answer | Back Directory | [Properties]
white or almost white crystalline granules or powder, odorless, slightly sweet. The sweetness of α-lactose is 15% of sucrose. The β-lactose has a higher sweetness than α-lactose. It is soluble in water, insoluble in alcohol, insoluble in ether and chloroform. The lactose monohydrate has a water content of 4.5% -5.5%, while the anhydrous hydrate has a water content of less than 1%. 9.75% (w / v) aqueous solution is isotonic with serum.
| [Stability and storage conditions]
It is easily contaminated by mold in moist conditions (humidity greater than 80%). The color of lactose may turn brown as storage time is extended with moist and heat accelerating this change. Alpha-lactose monohydrate is stable in air and is unaffected by moisture at room temperature, but amorphous form of lactose is dependent on the degree of dryness and may be subject to moisture to be converted to monohydrate. Monohydrate, when heated to 120 ℃, can be converted into anhydrous. A saturated solution of beta-lactose may be precipitated into α-lactose crystals during placement, and the solution has a twisted optical rotation property. Lactose should be placed in airtight container and stored in a cool dry place.
| [Compatibility & incompatibility]
Lactose can reacted with primary amine compounds to produce a brown product; the amorphous lactose is more likely participated into this reaction than crystalline lactose. Lactose has incompatibility with amino acids, aminophylline, amphetamine and isoniazid. Prescription containing tartrate, citrate or acetate, etc., or alkaline lubricants can accelerate the discoloration reaction. | [Production process]
Use the product of producing cheese or casein - whey as raw material, remove the protein content, followed by concentration, cooling, lactose crystallization and some other processes to obtain the dairy products. Mainly used in food industry, medicine and chemical reagents. Production process: raw whey material- → protein separation - →whey concentration - → cooling and crystallization of lactose - → molasses separation - → lactose washing - → drying (crude lactose) → refining treatment (refined lactose). Raw whey contains whey protein and a small amount of casein, which can be separated through adding line milk to the whey to pH 6.2 ~ 6.5 and simultaneously heating to over 90 ℃ for protein precipitation. Next, the cleared whey is then vacuumed concentrated to 35Be '(70% dry matter, Be' denotes Baume, a specific gravity unit) and cooled to 18-20 ° C for 24 hours to crystallize the lactose. In order to speed up the crystallization process and increase the yield, an appropriate amount of crystallization inducer (lactose powder or crystallized liquid lactose) can be added at the initial stage of crystallization.
Crystal should be separated out of molasses using centrifugal dewatering machine and washed with water and finally dried to obtain crude lactose. In the dairy industry, refined lactose is usually produced, but since the crude lactose is yellow and contains more impurities, it needs to be refined before obtaining refined lactose. First, dissolve the crude lactose in warm water; add the decolorant (bone charcoal or activated charcoal) to decolorize; or use ion exchange resin for salt treatment; or use milk protein and calcium chloride for further removing the proteins. Then again apply concentration, drying to make refined lactose. The protein precipitate separated during the production of lactose can be used as a feed, and the separated molasses can also be used as a raw material for feed or lactic acid fermentation.
| [Lactose intolerance]
Lactose digestion and malabsorption could be caused by congenital or secondary lactase deficiency. Lactase (β-galactosidase) catalyzes the hydrolysis of lactose to galactose and glucose, playing an important role in the digestion and absorption of lactose. Most babies contain high amount and high-activity intestinal lactase, being capable of fully digesting a large number of lactose in the diet. At about age 16, intestinal lactase begins to degenerate, and can finally lead to varying degrees of lactase deficiency in adults, which varies according their race. Infant lactase deficiency is mainly due to congenital or hereditary such as congenital intestinal mucosal abnormalities caused by lactase secretion dysfunction; can also be secondary to intestinal diseases such as Crohn's disease, ulcerative colitis, the virus Enteritis causes intestinal mucosa damage. Lactose intolerance can be prevented by removing or avoiding the presence of lactose in the diet. Infants can be feed with lactose-free formula while children can take well-fermented yogurt (in which the lactose has been converted by lactobacillus into lactic acid); adults should avoid drinking a lot of milk with empty stomach. Instead, you can first have a certain amount of other foods to reduce the relative concentration of lactose in the intestine so that bacteria are capable of decomposing it slowly with decomposition products being gradually absorbed.
|
|
|