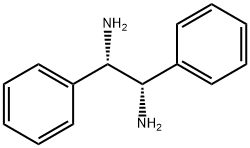
(1S,2S)-(-)-1,2-Diphenyl-1,2-ethanediamine synthesis
- Product Name:(1S,2S)-(-)-1,2-Diphenyl-1,2-ethanediamine
- CAS Number:29841-69-8
- Molecular formula:C14H16N2
- Molecular Weight:212.29
Yield:35132-20-8 80%
Reaction Conditions:
Stage #1: benzylidenaminewith (5R,5'R)-4,4,4',4'-tetrakis(2,5-dimethylphenyl)-5,5'-diphenyl-2,2'-bi(1,3,2-dioxaborolane) in tetrahydrofuran;methanol at 20 - 30;Schlenk technique;Inert atmosphere;Sealed tube;
Stage #2: with hydrogenchloride in methanol;water; pH=Ca. 3;Catalytic behavior;Reagent/catalyst;Solvent;
Steps:
3 Example 3
General procedure: In a clean and dry shlenk tube, add benzaldehyde (0.2mmol, 1.0eq),The system was pumped with nitrogen three times and protected by nitrogen. Under the protection of nitrogen, ammonia methanol solution (7.0M, 3.0mmol, 15.0eq) was added, and tetrahydrofuran (2mL) was re-evaporated with solvent, and the system was sealed.The reaction was stirred at room temperature for 1h, and under the protection of nitrogen, {System 1: [Diol diol(1-8)(0.08mmol, 0.4eq) and B2(neo)2(0.15mmol, 0.75eq)], or,System 2: [DB(1-4)(0.11mmol, 0.55eq)]}, stirring at room temperature (25-30°C) for 12-24h. Rotate the system to remove excess ammonia, add 1mL methanol, add hydrochloric acid (3.0M, 1mL)Adjust the pH to about 3, wash with dichloromethane several times,Add sodium hydroxide solution to the water phase to adjust the pH to about 12, extract with dichloromethane,The organic phases are combined, dried over anhydrous sodium sulfate, and the organic phase is spin-dried to obtain the product.
References:
CN111440205,2020,A Location in patent:Paragraph 0248-0252

100-52-7
951 suppliers
$20.37/250g

29841-69-8
355 suppliers
$6.00/1g
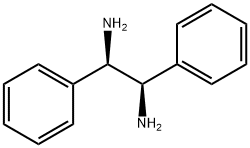
35132-20-8
361 suppliers
$7.00/1g
![Benzamide, N-[1,2-diphenyl-2-[(phenylmethyl)imino]ethyl]-](/CAS/20210305/GIF/1414364-68-3.gif)
1414364-68-3
0 suppliers
inquiry

29841-69-8
355 suppliers
$6.00/1g
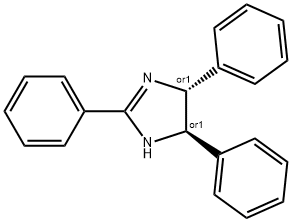
33722-46-2
3 suppliers
inquiry

29841-69-8
355 suppliers
$6.00/1g
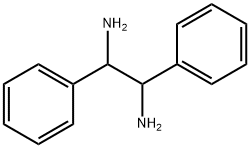
5700-60-7
80 suppliers
$207.03/10g

29841-69-8
355 suppliers
$6.00/1g

35132-20-8
361 suppliers
$7.00/1g
