4,6-Dichlorpyrimidin Chemische Eigenschaften,Einsatz,Produktion Methoden
R-S?tze Betriebsanweisung:
R34:Verursacht Ver?tzungen.
R20/21/22:Gesundheitssch?dlich beim Einatmen,Verschlucken und Berührung mit der Haut.
R36/37:Reizt die Augen und die Atmungsorgane.
S-S?tze Betriebsanweisung:
S26:Bei Berührung mit den Augen sofort gründlich mit Wasser abspülen und Arzt konsultieren.
S36/37/39:Bei der Arbeit geeignete Schutzkleidung,Schutzhandschuhe und Schutzbrille/Gesichtsschutz tragen.
S45:Bei Unfall oder Unwohlsein sofort Arzt zuziehen (wenn m?glich, dieses Etikett vorzeigen).
S27:Beschmutzte, getr?nkte Kleidung sofort ausziehen.
Chemische Eigenschaften
Yellow Solid
Verwenden
4,6-Dichloropyrimidine was used in the synthesis of
N-substituted azacalix[4]pyrimidines. It was used as starting reagent for the synthesis of disubstituted pyrimidines by tandem amination and Suzuki-Miyaura cross-coupling. It was also used in a biarylpyrimidine synthesis involving biaryl cross-coupling.
Allgemeine Beschreibung
Cyclic voltammograms of 4,6-dichloropyrimidine shows three cathodic waves, arising from sequential cleavage of carbon-chlorine bonds as well as the reduction of pyrimidine.
Synthese
The synthesis of 4,6-Dichloropyrimidine is as follows:Dissolve 105.5g of 4,6-diaminopyrimidine in 660.0g of 31% hydrochloric acid and pour into a 2000ml bottle to cool to -5 ° C and dropwise add 500.3g of 33% sodium nitrite. After 2 hours of reaction, HPLC detects 4,6-diamino Pyrimidine is less than 0.5%. 42.8 g of cuprous chloride and 214.0 g of 31% hydrochloric acid are prepared in a 2000 ml bottle. The diazonium salt mother liquor is added dropwise to the bottle. After the dropwise reaction, the reaction is performed at 45 ° C for 2 hours. .Extract with 400g of recovered trichloroethane (200x2 times less than new ones), combine the organic layers for distillation, control the water flush pump 5KPa, temperature 40-140 , collect 404.2g of solvent in the early 40-90 , 90-140 in the later The product was collected at a temperature of 14 ° C to obtain 146.9 g of 4,6-dichloropyrimidine. The yield was 86.4% (based on formazan hydrochloride) and the purity was 99.4%.
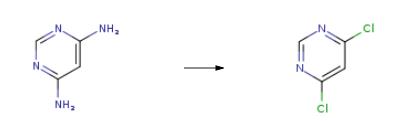
4,6-Dichlorpyrimidin Upstream-Materialien And Downstream Produkte
Upstream-Materialien
Downstream Produkte

