| Identification | Back Directory | [Name]
DEXCHLORPHENIRAMINE MALEATE | [CAS]
2438-32-6 | [Synonyms]
Polamin
Sensidyn
Phenamin
polaramin
Polaramine
d-Form maleate
poraminemaleate
polaraminemaleate
D-CHLORPHENIRAMINE MALEATE
Chlorpheniramine d-maleate
DEXCHLORPHENIRAMINE MALEATE
DEXCHLOROPHENIRAMINE MALEATE
(+)-Chloropromazine Maleate
dextrochlorpheniraminemaleate
s-(+)-chlorpheniraminemaleate
d-Chlorpheniramine·maleic acid
DEXCHLORPHENIRAMINEMALEATE,USP
(s)-(+)-chlorpheniraminemaleate
(+)-CHLORPHENIRAMINE MALEATE SALT
s-(+)-chlorpheniramine maleate salt
Dexchlorpheniramine hydrogen maleate
Dexchlorpheniramine Maleate (300 mg)
DexchlorpheniramineMaleate-Bp/Ep/Usp
γ-(4-Chlorophenyl)-N,N-dimethyl-2-pyridinepropanamine
g-(4-chlorophenyl)-N,N-dimethyl-2-pyridinepropanaminemaleate
2-[p-Chloro-α-[2-(diMethylaMino)ethyl]benzyl]pyridine Maleate
d-2-[p-Chloro-α-(2-dimethylaminoethyl)benzyl]pyridine bimaleate
(+)-2-(p-chloro-alpha-(2-(dimethylamino)ethyl)benzyl)pyridinemaleate
GAMMA-[4-CHLOROPHENYL]-N,N-DIMETHYL-2-PYRIDINEPROPANAMINE MALEATE SALT
(s)-γ-(4-chlorophenyl)-n,n-dimethyl-2-pyridinepropanamine maleate salt
pyridine,2-(p-chloro-alpha-(2-(dimethylamino)ethyl)benzyl)-,maleate(1:1),(+
(γS)-γ-(4-Chlorophenyl)-N,N-diMethyl-2-pyridinepropanaMine (2Z)-2-Butenedioate
[gamma-(4-chlorophenyl)-pyridine-2-propyl](dimethyl)ammonium hydrogen (Z)-maleate
Pyridine, 2-[p-chloro-α-[2-(dimethylamino)ethyl]benzyl]-, maleate (1:1), (S)- (8CI)
2-Pyridinepropanamine, γ-(4-chlorophenyl)-N,N-dimethyl-, (S)-, (Z)-2-butenedioate (1:1)
2-Pyridinepropanamine, γ-(4-chlorophenyl)-N,N-dimethyl-, (γS)-, (2Z)-2-butenedioate (1:1)
γ-(4-Chlorophenyl)-N,N-dimethyl-2-pyridinepropanamine, (+)-Chlorpheniramine maleate salt
2-Pyridinepropanamine, .gamma.-(4-chlorophenyl)-N,N-dimethyl-, (.gamma.S)-, (2Z)-2-butenedioate (1:1) | [EINECS(EC#)]
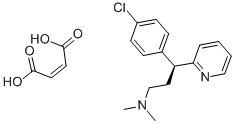
204-037-5 | [Molecular Formula]
C20H23ClN2O4 | [MDL Number]
MFCD00079046 | [MOL File]
2438-32-6.mol | [Molecular Weight]
390.86 |
| Chemical Properties | Back Directory | [Melting point ]
112-115 °C(lit.) | [alpha ]
D25 +44.3° (c = 1 in dimethylformamide) | [storage temp. ]
Refrigerator | [solubility ]
Very soluble in water, freely soluble in ethanol (96 per cent), in methanol and in methylene chloride. | [form ]
neat | [color ]
White to off-white | [PH]
pH(10g/l, 25℃) : 4.0~5.0 | [Merck ]
13,2198 |
| Hazard Information | Back Directory | [Chemical Properties]
White Solid | [Uses]
bone resorption inhibitor, Antihistaminic. | [Uses]
The S-enantiomer of Chlorpheniramine (C424300). Antihistaminic. | [Description]
Dexchlorpheniramine Maleate is the maleate salt form of dexchlorpheniramine, an alkylamine, and first-generation histamine antagonist with anti-allergic activity. Dexchlorpheniramine maleate competitively blocks H1 receptors, thereby preventing the actions of histamine on bronchial smooth muscle, capillaries and gastrointestinal (GI) smooth muscle. This prevents histamine-induced bronchoconstriction, vasodilation, increased capillary permeability, and GI smooth muscle spasms. | [Originator]
Polaramine,Schering,US,1958 | [Definition]
ChEBI: Dexchlorpheniramine maleate is an organic molecular entity. | [Manufacturing Process]
Twenty grams of d-phenylsuccinic acid and 28 grams of 3-(2-pyridyl)-3-pchlorophenyl-N,N-dimethylpropylamine are dissolved in 400 ml of absolute
ethyl alcohol and allowed to stand at room temperature until crystallization is
effected. The crystals are filtered, washed with absolute ethyl alcohol and
recrystallized from 300 ml of this solvent in the same manner. The crystals
are recrystallized twice from 80% ethyl alcohol using 3.5 ml per gram of
compound in the manner described above and pure d-3-(2-pyridyl)-3-pchlorophenyl-N,Ndimethylpropylamine-d-phenylsuccinate is obtained, melting
point 145-147°C.
This salt is shaken with 100 ml of diethyl ether and 50 ml of 20% aqueous
potassium carbonate; the ether layer is separated, dried over anhydrous
potassium carbonate, filtered and the ether is removed in vacuo. The d-3-(2-
pyridyl)-3-p-chlorophenyl-N,N-dimethylpropylamine so obtained is a mobile
oil.
4.3 grams of the above base and 1.8 grams of maleic acid are dissolved in 20
ml isopropyl acetate and kept at room temperature until crystallization is
complete. The crystals are filtered, washed with ethyl acetate and
recrystallized from 15 ml of this solvent in the same manner. The crystalline
d-3-(2-pyridyl)-3-p-chlorophenyl-N,N-dimethylpropylamine maleate so formed
is then filtered off and dried. MP 113-115°C from US Patent 3,030,371. | [Brand name]
Mylaramine (Morton
Grove); Polaramine (Schering). | [Therapeutic Function]
Antihistaminic | [General Description]
Dexchlorpheniramine(Polaramine) is the dextrorotatory enantiomer of chlorpheniramine.In vitro and in vivo studies of the enantiomers ofchlorpheniramine showed that the antihistaminic activity existspredominantly in the dextro isomer. As mentioned previously,the dextro isomer has the (S) configuration, which issuperimposable on the (S) configuration of the more activelevorotatory enantiomer of carbinoxamine. | [Biochem/physiol Actions]
H1 histamine receptor antagonist; active isomer.Chlorpheniramine maleate is clinically used as a topical ointment to treat skin disorders such as sunburn, urticaria, angioedema, pruritus and insect bites. | [Mode of action]
Dexchlorpheniramine, the d-isomer of the racemic compound chlorpheniramine, is two times more active than chlorpheniramine. Dexchlorpheniramine does not prevent the release of histamine, but rather, competes with free histamine for binding at the H1-receptor sites, and competitively antagonizes the effects of histamine on H1- receptors in the GI tract, uterus, large blood vessels, and bronchial muscle. Blockade of H1-receptors also suppresses the formation of oedema, flare, and pruritus that result from histaminic activity. Since dexchlorpheniramine binds to central and peripheral H1-receptors, sedative effects are likely to occur. H1- antagonists are structurally similar to anticholinergic agents and therefore possess the potential to exhibit anticholinergic properties of varying degrees. They also have antipruritic effects. Dexchlorpheniramine has high antihistaminic activity, moderate anticholinergic effects and minimal sedative effects. The medicine does not possess antiemetic properties. | [References]
[1] P. E. CHAVES. Assessment of cytotoxicity, genotoxicity, and mutagenicity of the dexchlorpheniramine reference standard and pharmaceutical formula in human peripheral blood mononuclear cells[J]. Brazilian Journal of Pharmaceutical Sciences, 2022, 18 1. DOI:10.1590/s2175-97902022e20096.
[2] GUSTAVO FACCHINI MSC. Ultraviolet A photosensitivity profile of dexchlorpheniramine maleate and promethazine-based creams: Anti-inflammatory, antihistaminic, and skin barrier protection properties[J]. Journal of Cosmetic Dermatology, 2017, 16 4: e59-e67. DOI:10.1111/jocd.12349.
[3] MASAAKI TAGAWA. Neuroimaging of histamine H1-receptor occupancy in human brain by positron emission tomography (PET): a comparative study of ebastine, a second-generation antihistamine, and (+)-chlorpheniramine, a classical antihistamine.[J]. British journal of clinical pharmacology, 2001, 52 5 1: 501-509. DOI:10.1046/J.1365-2125.2001.01471.X.
[4] DONG LIANG. Identification of anthelmintic parbendazole as a therapeutic molecule for HNSCC through connectivity map-based drug repositioning[J]. Acta Pharmaceutica Sinica B, 2022, 12 5: Pages 2429-2442. DOI:10.1016/j.apsb.2021.12.005. |
|
| Company Name: |
AKASH PHARMA EXPORTS
|
| Tel: |
+91-9388123451 +91-9846039283 |
| Website: |
www.UNDERCONSTRUCTION |
|