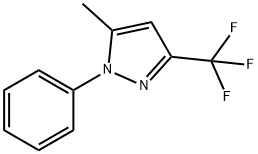
3-TRIFLUOROMETHYL-5-METHYL-1-(PHENYL)PYRAZOLE synthesis
- Product Name:3-TRIFLUOROMETHYL-5-METHYL-1-(PHENYL)PYRAZOLE
- CAS Number:111079-04-0
- Molecular formula:C11H9F3N2
- Molecular Weight:226.2
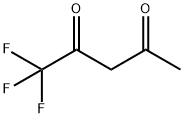
367-57-7
287 suppliers
$10.00/2g
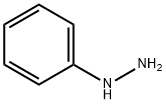
100-63-0
362 suppliers
$10.00/1g
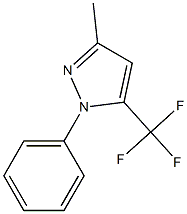
129585-50-8
0 suppliers
inquiry

111079-04-0
17 suppliers
inquiry
Yield:-
Reaction Conditions:
Stage #1: 1,1,1-Trifluoro-2,4-pentanedione;phenylhydrazinewith acetic acid at 7 - 20; for 2 h;
Stage #2: with pyrographite
Steps:
6.A EXAMPLE 6; Step A: Preparation of 2-Methyl-1-phenyl-4-(trifuoromethyl)-1H-pyrazole
A solution of 1,1,1-trifluoropentane-2,4-dione (20.0 g, 0.130 mole) in glacial acetic acid (60 mL) was cooled to 7° C. using an ice/water bath. Phenylhydrazine (14.1 g, 0.130 mole) was added dropwise over a period of 60 minutes. The reaction mass temperature increased to 15° C. during the addition. The resulting orange solution was held under ambient conditions for 60 minutes. The bulk of the acetic acid was removed by stripping on a rotary evaporator at a bath temperature of 65° C. The residue was dissolved in methylene chloride (150 mL). The solution was washed with aqueous sodium bicarbonate (3 g in 50 mL water). The purple-red organic layer was separated, treated with activated charcoal (2 g) and MgSO4, then filtered. Volatiles were removed on a rotary evaporator. The crude product consisted of 28.0 g of a rose-colored oil, which contained 89% the desired product and 11% 1-phenyl-5-(trifluoromethyl)-3-methylpyrazole. [0321] 1H NMR (DMSO-D6) 67 2.35 (s,3H), 6.76 (s,1H), 7.6-7.5 (m,5H).
References:
US2003/229050,2003,A1 Location in patent:Page 27

100-63-0
362 suppliers
$10.00/1g

111079-04-0
17 suppliers
inquiry
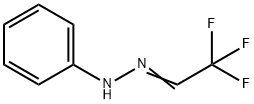
81963-42-0
0 suppliers
inquiry

111079-04-0
17 suppliers
inquiry

367-57-7
287 suppliers
$10.00/2g

100-63-0
362 suppliers
$10.00/1g

111079-04-0
17 suppliers
inquiry
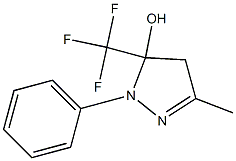
109954-11-2
0 suppliers
inquiry