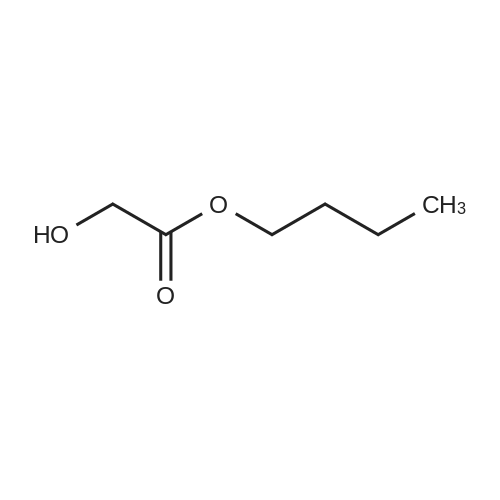
| 91.5% |
at 20℃; |
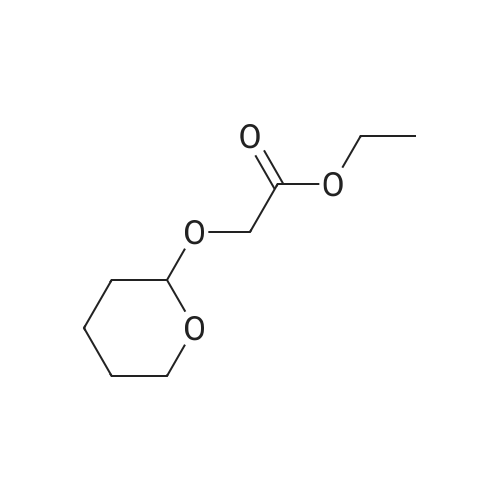
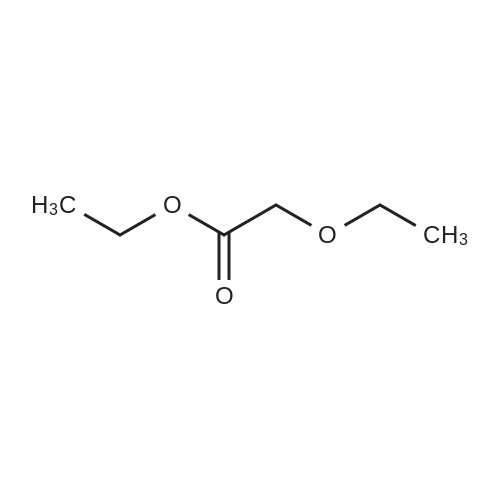
(1) Preparation of tetrahydropyran-2-yloxy-acetic acid ethyl ester (Formula 28, Scheme 4)[0378] To a stirred solution of ethyl glycolate (35.3g, 0.339mol) containing a few crystals of />toluene sulfonic acid, 3,4-dihydropyran (30.Og, 0.357mol) was added1 1 <n="113"/>dropwise (15g over one hour followed by 15g over 30min). After stirring overnight at room temperature, the mixture was diluted with diethyl ether (8OmL) and washed with a NaHCO3 solution (from 3OmL sat. NaHCO3 and 1OmL water). The organic layer was separated and dried (Na2SO4) followed by evaporation of the ether. The residue was distilled under high vacuum to give 58.4g (91.5percent) of 28 as a clear liquid. 1H NMR (CDCL3, 400MHz)δ: 1.29 (t, 3H, J = 7.1 Hz, CH3), 1.53 -1.95 (m, 6H, 3,4,5-THP-CH2's), 3.50-3.55 (m, IH, 6-THP-CH2), 3.83 - 3.89 (m, IH, 6-THP-CH2), 4.19 (s, 2H, OCH2CO2R), 4.20 (t, 2H, J = 7.1 Hz, CH2Me), 4.75 (m, IH, THP-CH). 13C NMR (CDCl3, 100MHz) δ: 14.1, 18.7, 25.2, 30.0, 60.7, 61.9, 63.8, 170.4. |
| 81% |
With pyridinium p-toluenesulfonate In dichloromethane |
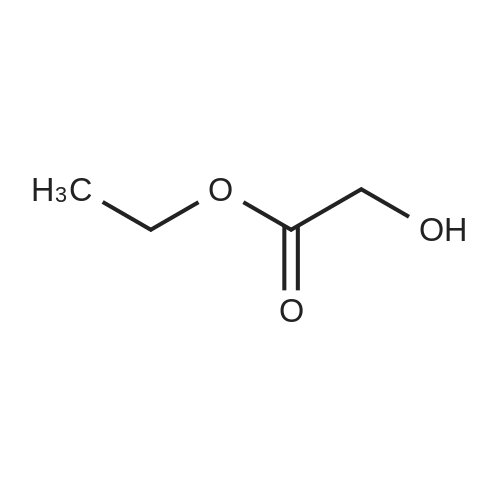
Step 1) ethyl 2-(tetrahydro-2H-pyran-2-yloxy)acetate
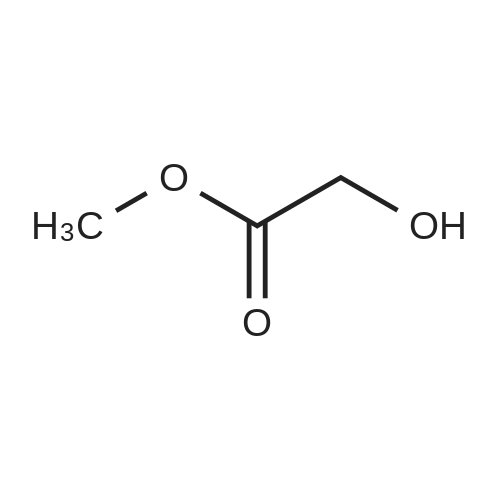
To a mixture of ethyl 2-hydroxyacetate (2 g, 20 mmol, Aldrich) and 3,4-dihydro-2H-pyran (3.2 g, 40 mmol, Alfa) in 40 mL of CH2Cl2 was added PPTS (500 mg, 2 mmol) slowly at rt.
The mixture was stirred at rt for 4 hours, and then the mixture was washed with brine (20 mL*2), the combined organic phases were dried over Na2SO4 and concentrated in vacuo.
The residue was purified by a silica gel column chromatography (PE: EtOAc=20:1) to give colorless oil (3.01 g, 81percent).
1H NMR (400 MHz, CDCl3): δ 1.22-1.38 (m, 4H), 1.55-1.63 (m, 3H), 1.69-1.88 (m, 3H), 3.50-3.53 (m, 1H), 3.82-3.88 (m, 1H), 4.18-4.23 (m, 4H), 4.73-4.74 (t, J=4 Hz, 1H).
|
| 81% |
With pyridinium p-toluenesulfonate In dichloromethane at 20℃; for 4 h; |
To a mixture of ethyl 2-hydroxyacetate (2 g, 20 mmol, Aldrich) and 3,4-dihydro-2H-pyran (3.2 g, 40 mmol, Alfa) in 40 mL of CH2Cl2 was added PPTS (500 mg, 2 mmol) slowly at rt.
The mixture was stirred at rt for 4 hours, and then the mixture was washed with brine (20 mLx2), the combined organic phases were dried over Na2SO4 and concentrated in vacuo.
The residure was purified by a silica gel column chromatography (PE: EtOAc =20:1) to give colorless oil (3.01 g, 81 percent).
1H NMR (400MHz, CDCl3): δ 1.22 - 1.38 (m, 4H), 1.55 - 1.63 (m, 3H), 1.69 - 1.88 (m, 3H), 3.50 - 3.53 (m, 1H), 3.82 - 3.88 (m, 1H), 4.18 - 4.23 (m, 4H), 4.73 - 4.74 (t, J=4Hz, 1H). |
| 81% |
With pyridinium p-toluenesulfonate In dichloromethane at 20℃; for 4 h; Inert atmosphere |
To a mixture of ethyl 2 -hydroxy acetate (2 g, 20 mmol, TCI) and 3,4- dihydro-2H-pyran (3.2 g, 40 mmol, Alfa) in 40 mL Of CH2Cl2 was added PPTS (500 mg, 2 mmol, Aldrich) in portions at rt. The mixture was stirred at rt for 4 hours. The reaction mixture was then washed with brine, and the organic layer was separated and the combined organic phases were dried over Na2SO4, concentrated in vacuo. The residue was purified by a silica gel column chromatography (20: 1 (v/v) petroleum ether / EtOAc) to give the desired compound as colorless oil (3.01 g, 81 percent ).1H NMR (400MHz, CDCl3): δ 1.25 - 1.32 (m, 3H), 1.55 - 1.63 (m, 3H), 1.69 - 1.88 (m, 3H), 3.50 - 3.53 (m, IH), 3.82 - 3.88 (m, IH), 4.18 - 4.23 (m, 4H), 4.73 (t, J=3.2Hz, IH). |
| 58% |
With toluene-4-sulfonic acid In toluene at 20℃; |
Step 1
Preparation of ethyl 2-(tetrahydro-2H-pyran-2-yloxy)acetate
A mixture of 3,4-dihydro-2H-pyran (20.4 g, 242.3 mmol), ethyl 2-hydroxyacetate (24.0 g, 230.8 mmol) and TsOH (0.794 g, 4.6 mmol) in toluene (150 mL) was stirred at room temperature overnight.
The resulting mixture was concentrated in vacuo and the residue was purified by column chromatography on silica gel (2percent EtOAc in petroleum ether) to afford ethyl 2-(tetrahydro-2H-pyran-2-yloxy)acetate (25.2 g, 58percent) as a colorless oil. |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 120K+ Compounds
120K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping