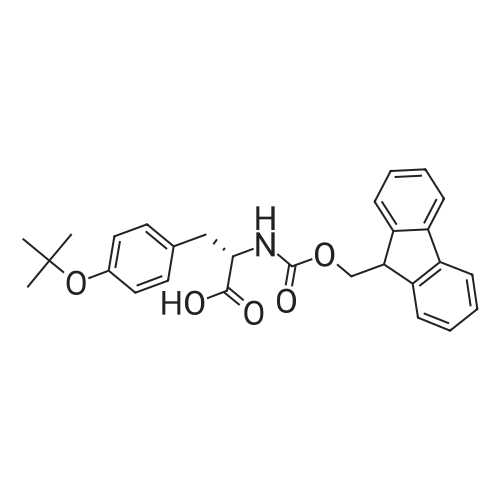
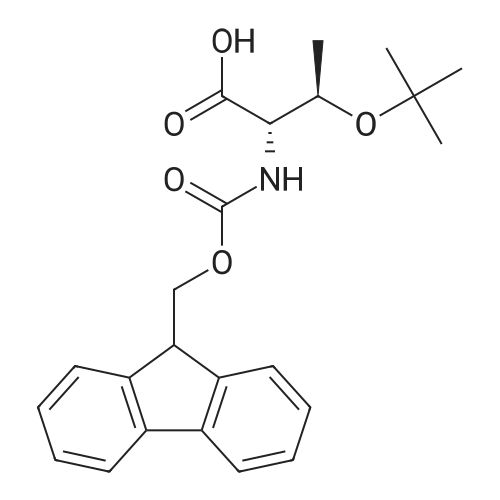
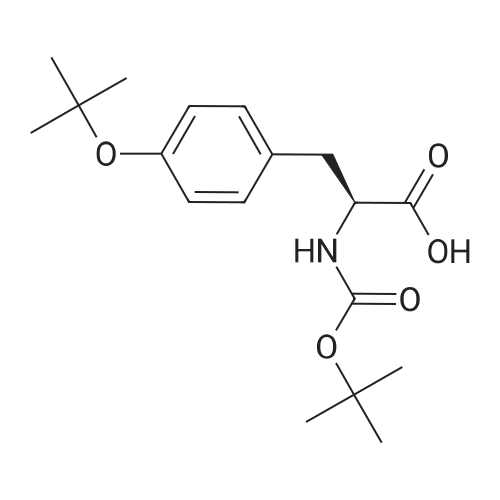
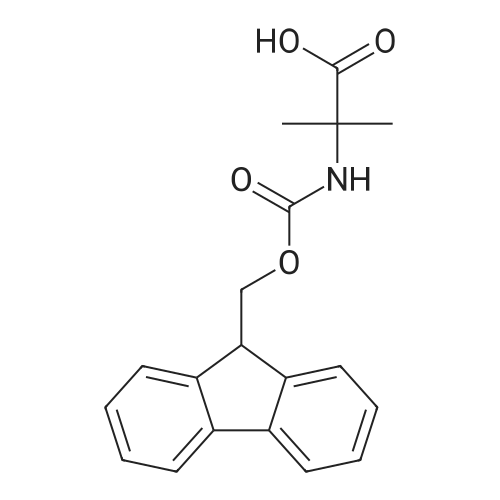
| 98% |
|
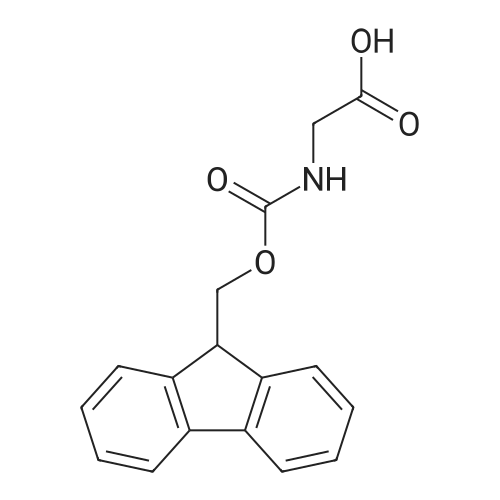
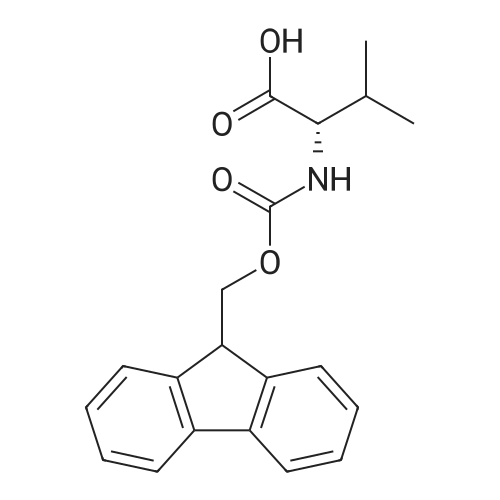
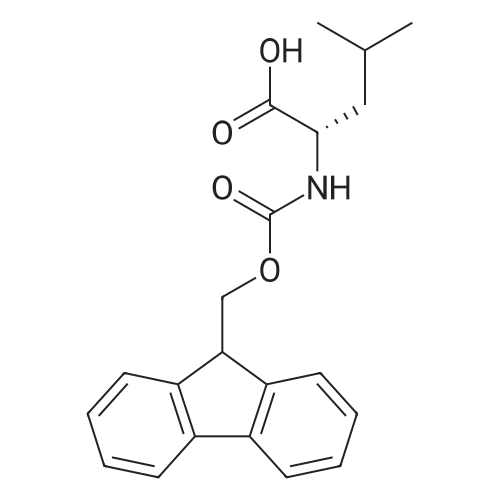
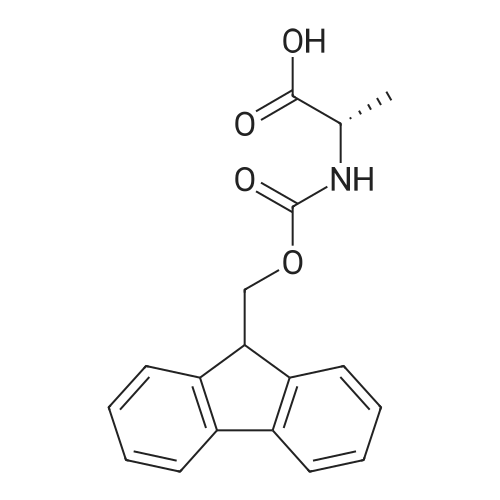
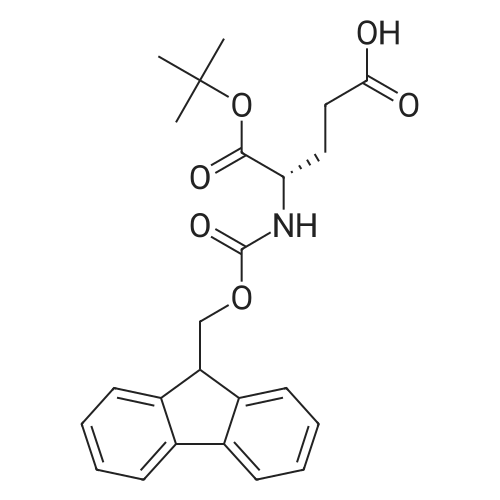
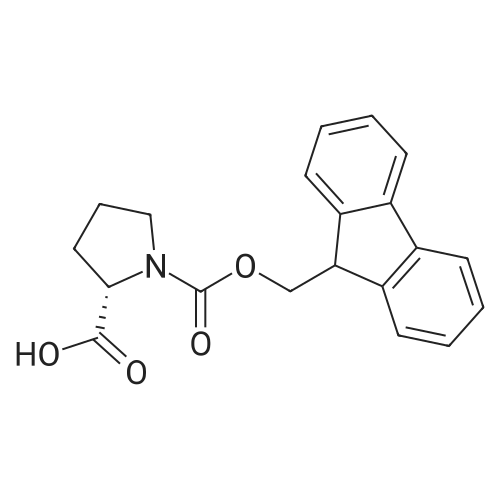
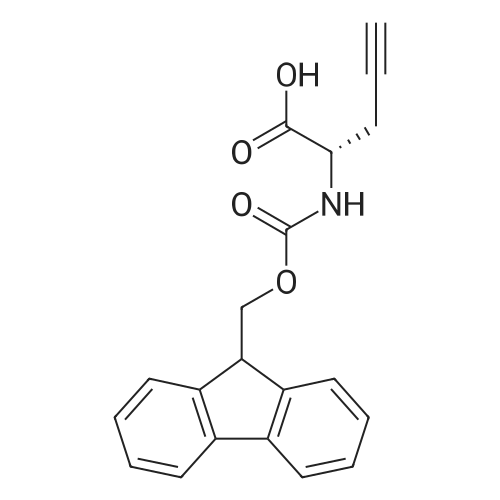
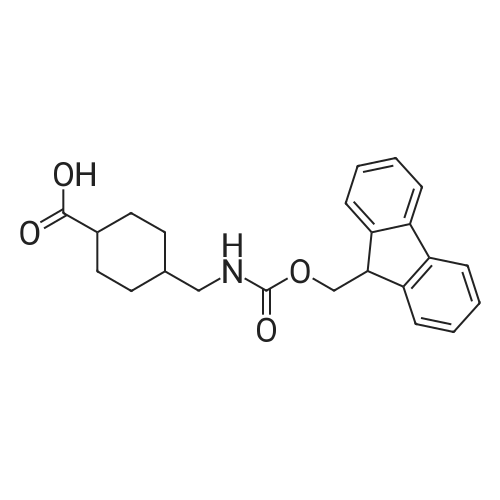
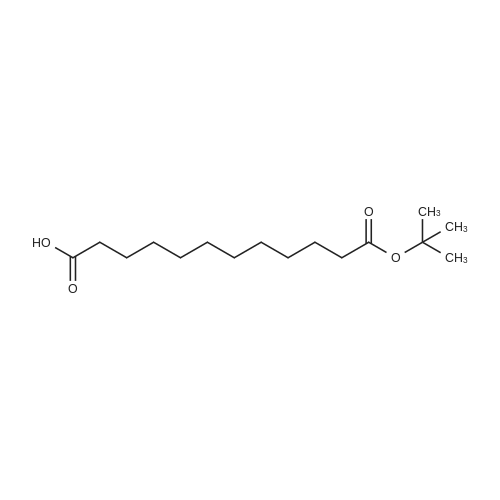
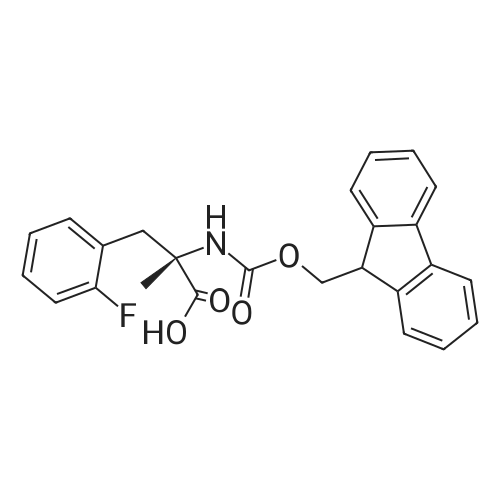
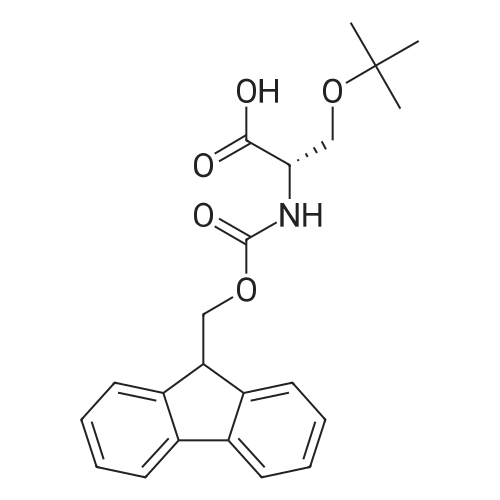
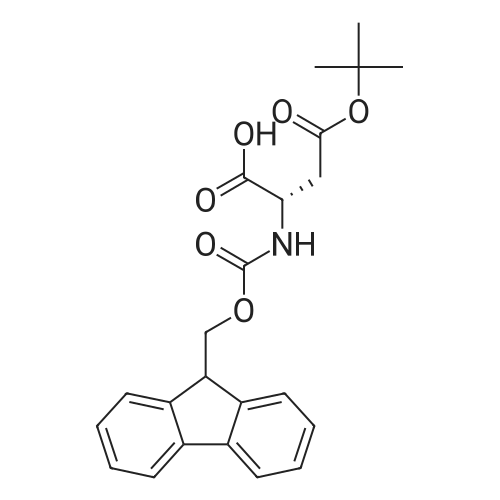
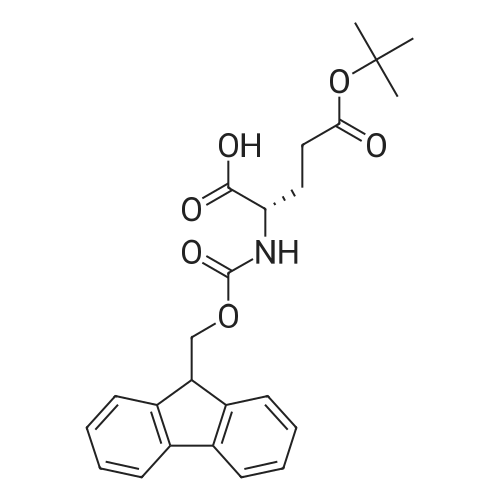
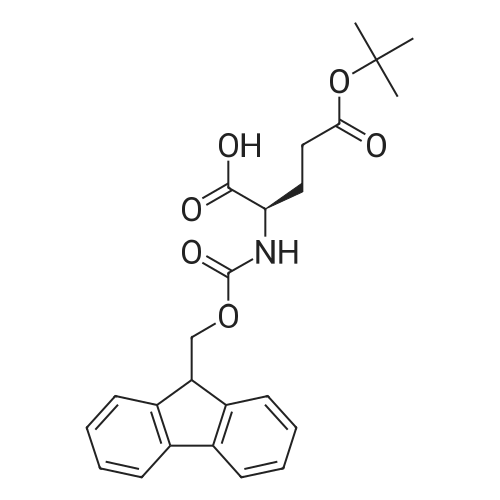
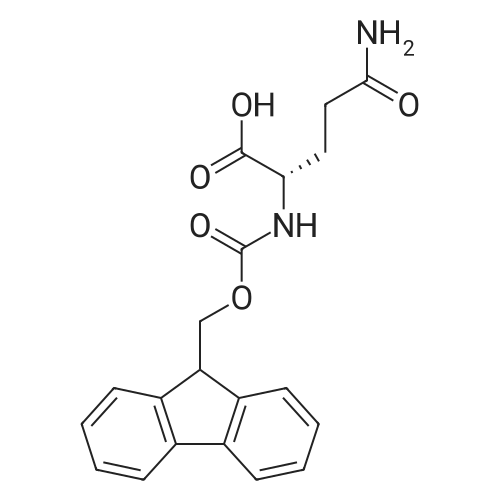
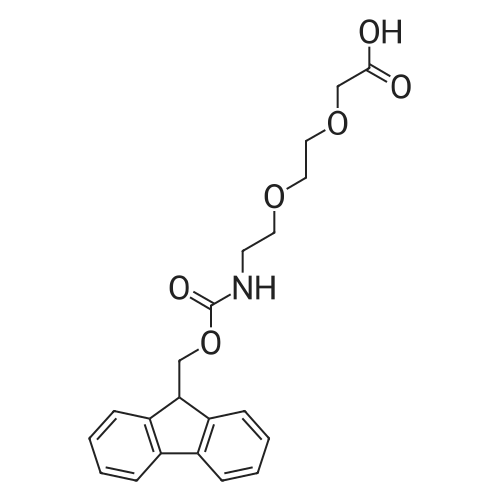
Synthetic protocol: Wang Fmoc-Lys(Mtt) resin 0.26 mmol/g (1, 11.2 g, 2.90 mmol) was left to swell in dichloromethane (100 ml) for 45 minutes. Fmoc group was removed by treatment with 20% piperidine in N,N-dimethylformamide (1 x 5 min, 1 x 10 min, 1 x 30 min, 3 x 100 15 ml). Resin was washed with N,N-dimethylformamide (3 x 90 ml), 2-propanol (3 x 90 ml) and dichloromethane (3 x 90 ml). A solution of {2-[2-(9H-fluoren-9ylmethoxycarbonylamino)-ethoxy]-ethoxy}-acetic acid (Fmoc-OEG-OH, 2.23 g, 5.80 mmol), 0-(6-chlorobenzotriazol-1-yi)-N,N,N',N'-tetramethyluronium tetrafluoroborate (TCTU, 2.06 g, 5.80 mmol) and N,N-diisopropylethylamine (2.02 ml, 11.6 mmol) in N,N20 dimethylformamide (100 ml) was added to resin and the mixture was shaken for 1 hour. Resin was filtered and washed with N,N-dimethylformamide (3 x 90 ml), dichloromethane (3 x 90 ml) and N,N-dimethylformamide (3 x 90 ml). Fmoc group was removed by treatment with 20% piperidine in N,N-dimethylformamide (1 x 5 min, 1 x 10 min, 1 x 30 min, 3 x 100 ml). Resin was washed with N,N-dimethylformamide (3 x 90 25 ml), 2-propanol (3 x 90 ml) and dichloromethane (3 x 90 ml). Solution of {2-[2-(9Hfluoren-9-ylmethoxycarbonylamino)-ethoxy]-ethoxy}-acetic acid (Fmoc-OEG-OH, 2.23 g, 5.80 mmol), 0-(6-chloro-benzotriazol-1-yi)-N,N,N',N'-tetramethyluronium tetrafluoroborate (TCTU, 2.06 g, 5.80 mmol) and N,N-diisopropylethylamine (2.02 ml, 11.6 mmol) in N,N-dimethylformamide (100 ml) was added to resin and mixture was 30 shaken for 1.5 hour. Resin was filtered and washed with N,N-dimethylformamide (3 x 90ml), dichloromethane (3 x 90 ml) and N,N-dimethylformamide (3 x 90 ml). Fmoc group was removed by treatment with 20% piperidine in N,N-dimethylformamide (1 x 5 min, 1 x 10 min, 1 x 30 min, 3 x 100 ml). Resin was washed with N,N-dimethylformamide (3 x 90 ml), 2-propanol (3 x 90 ml) and dichloromethane (3 x 90 ml). Solution of (S)-2-(9H5 fluoren-9-ylmethoxycarbonylamino)-pentanedioic acid 1-tert-butyl ester (Fmoc-LGiuOtBu, 1.85 g, 4.35 mmol), 0-(6-chloro-benzotriazol-1-yi)-N,N,N',N'-tetramethyluronium tetrafluoroborate (TCTU, 1.55 g, 4.35 mmol) and N,N-diisopropylethylamine (1.36 ml, 7.82 mmol) in N,N-dimethylformamide (100 ml) was added to resin and mixture was shaken for 1.5 hour. Resin was filtered and washed with N,N-dimethylformamide (3 x 90 10 ml), dichloromethane (3 x 90ml) and N,N-dimethylformamide (3 x 90 ml). Fmoc group was removed by treatment with 20% piperidine in N,N-dimethylformamide (1 x 5 min, 1 x 10 min, 1 x 30 min, 3 x 100 ml). Resin was washed with N,N-dimethylformamide (3 x 90 ml), 2-propanol (3 x 90 ml) and dichloromethane (3 x 90 ml). Solution of <strong>[188715-40-4]4-[(9H-fluoren-9-ylmethoxycarbonylamino)methyl]cyclohexanecarboxylic acid</strong> 15 (<strong>[188715-40-4]Fmoc-Trx-OH</strong>, 1.65 g, 4.35 mmol), 0-(6-chloro-benzotriazol-1-yi)-N,N,N',N'tetramethyluronium tetrafluoroborate (TCTU, 1.55 g, 4.35 mmol) and N,Ndiisopropylethylamine (1.36 ml, 7.82 mmol) in N,N-dimethylformamide (100 ml) was added to resin and mixture was shaken for 2 hours. Resin was filtered and washed with N,N-dimethylformamide (3 x 90 ml), dichloromethane (3 x 90ml) and N,N20 dimethylformamide (3 x 90 ml). Fmoc group was removed by treatment with 20% piperidine in N,N-dimethylformamide (1 x 5 min, 1 x 10 min, 1 x 30 min, 3 x 100 ml). Resin was washed with N,N-dimethylformamide (3 x 90 ml), 2-propanol (3 x 90 ml) and dichloromethane (3 x 90 ml). Solution of icosanedioic acid mono-tert-butyl ester (C20(0tBu)-OH, 1. 73 g, 4.35 mmol), 0-(6-chloro-benzotriazol-1-yi)-N,N,N',N'25 tetramethyluronium tetrafluoroborate (TCTU, 1.55 g, 4.35 mmol) and N,Ndiisopropylethylamine (1.36 ml, 7.82 mmol) in N,N-dimethylformamide (100 ml) was added to resin and mixture was shaken for 2 hours. Resin was filtered and washed with N,N-dimethylformamide (3 x 90 ml), dichloromethane (3 x 90 ml), N,Ndimethylformamide (3 x 90 ml) and dichloromethane (3 x 90 ml). Mtt group was 30 removed by treatment with 80% 1,1,1,3,3,3-hexafluoro-2-propanol in dichloromethane (2 x 10 min, 2 x 30 min, 4 x 100 ml). Resin was washed with dichloromethane (6 x 90 ml) and N,N-dimethylformamide (3 x 90 ml). Solution of bromoacetic acid (8.06 g, 58.0 mmol) and N,N '-diisopropylcarbodiimide (DIC, 7.60 ml, 49.3 mmol) in N,Ndimethylformamide (100 ml) was added to resin and mixture was shaken for 40 35 minutes. Resin was filtered and washed with N,N-dimethylformamide (5 x 90 ml) and dichloromethane (12 x 90 ml). The product was cleaved from resin by treatment with trifluoroacetic acid (100 ml) for 1 hour. Resin was filtered off and washed with trifluoroacetic acid (1 x 50 ml) and dichloromethane (7 x 70 ml). Solutions were combined and solvents were evaporated to dryness giving a thick brownish oil. Yield: 3.28 g (98%). 5 1H NMR spectrum (300 MHz, Ac0D-d4, 80 C, dH): 4.68 (dd, J=8.0 and 5.4 Hz, 1 H); 4.60 (dd, J=7.9 and 5.3 Hz, 1 H); 4.16 (s, 2 H); 4.12 (s, 2 H); 3.94 (s, 2 H); 3.81-3.61 (m, 12 H); 3.59-3.44 (m, 4 H); 3.32 (t, J=6.8 Hz, 2 H); 3.14 (d, J=6.8 Hz, 2 H); 2.491.79 (m, 15 H)... |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 120K+ Compounds
120K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping


 Deacetylase Activity.jpg)