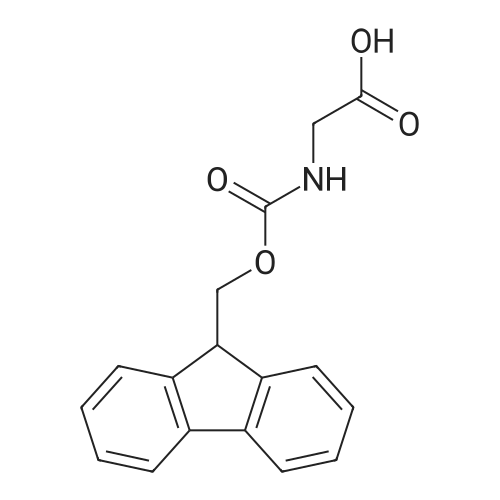
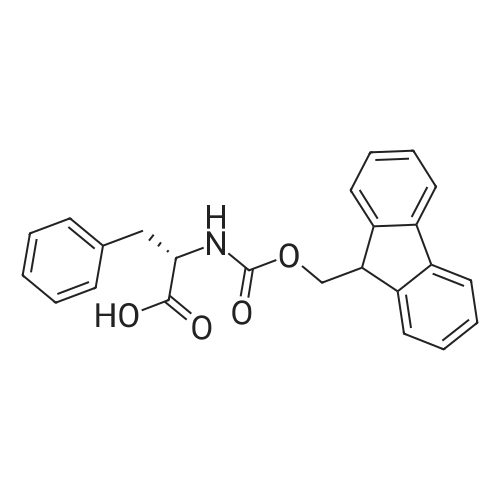
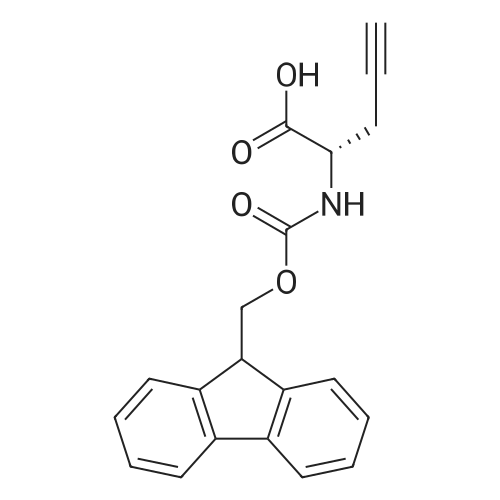
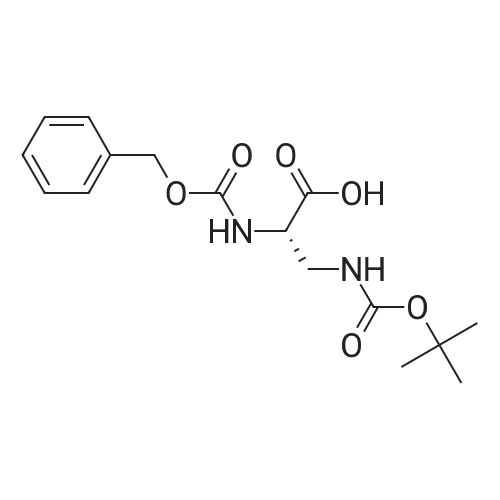
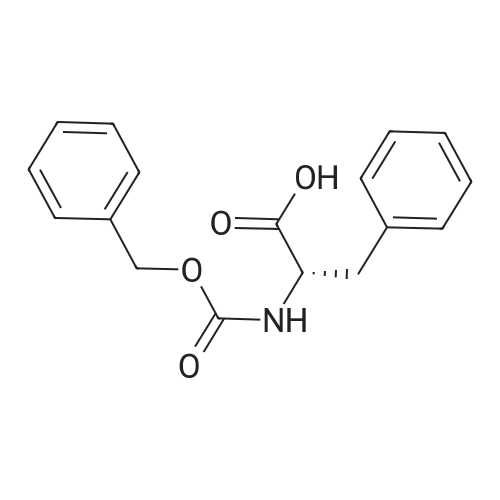
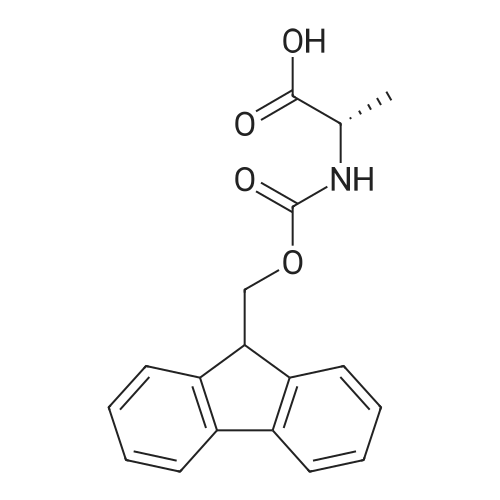
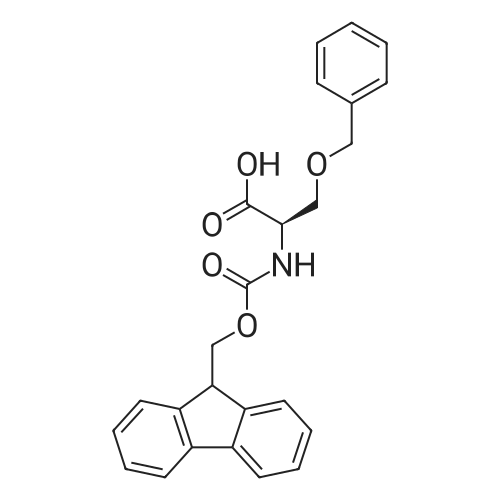
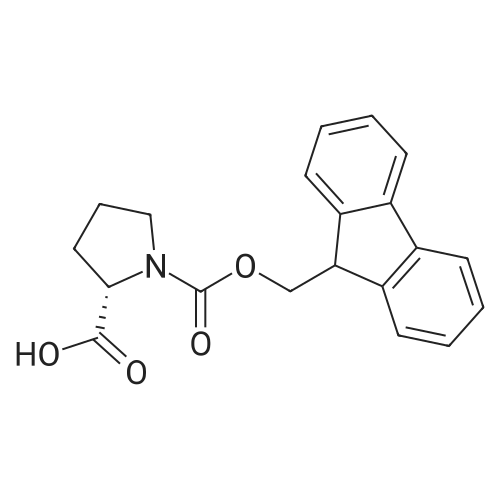
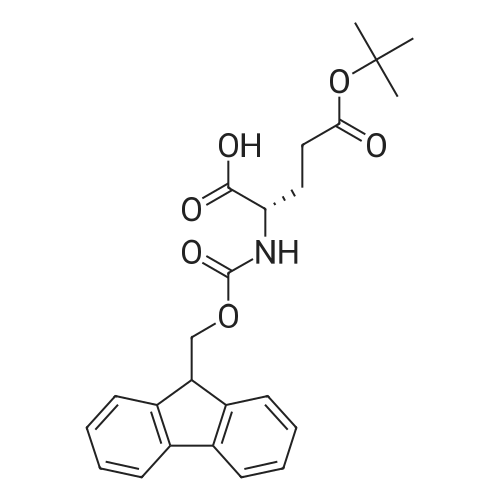
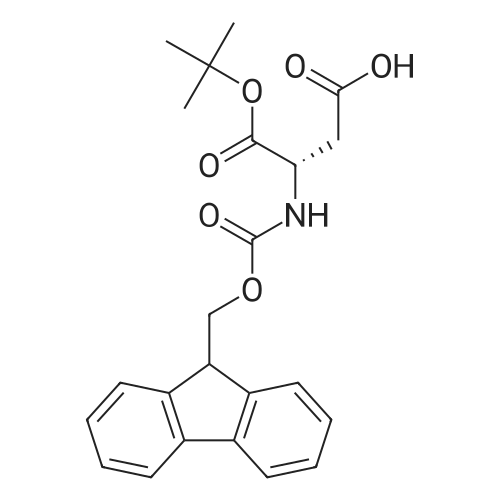
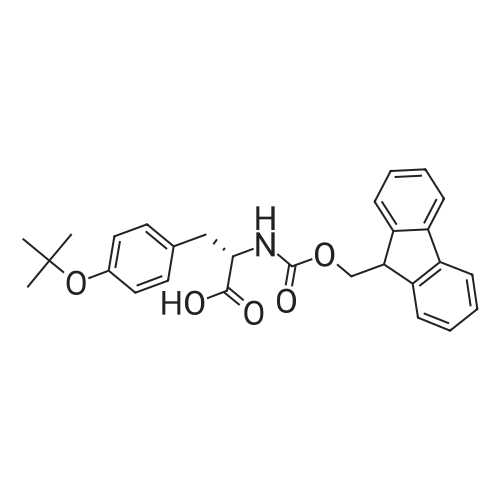
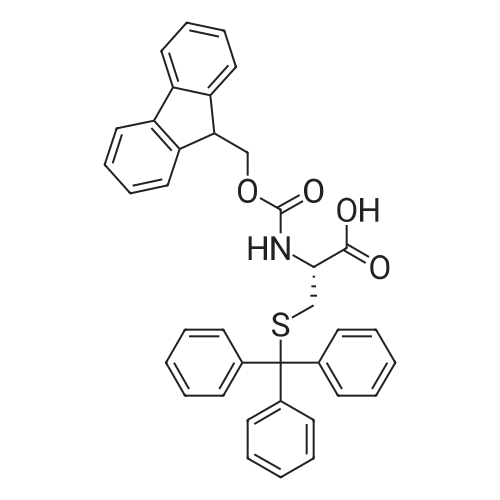
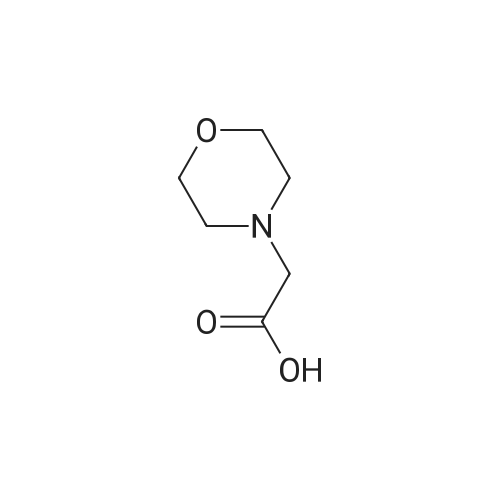
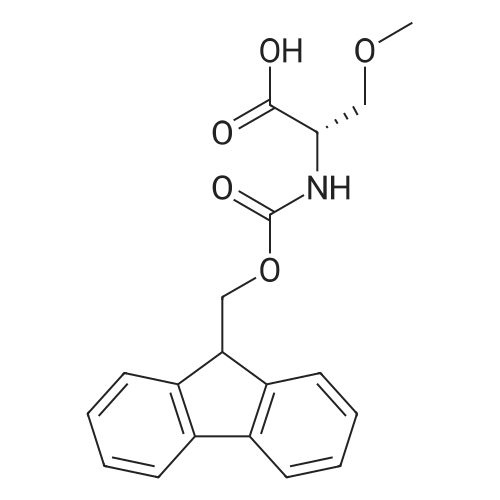
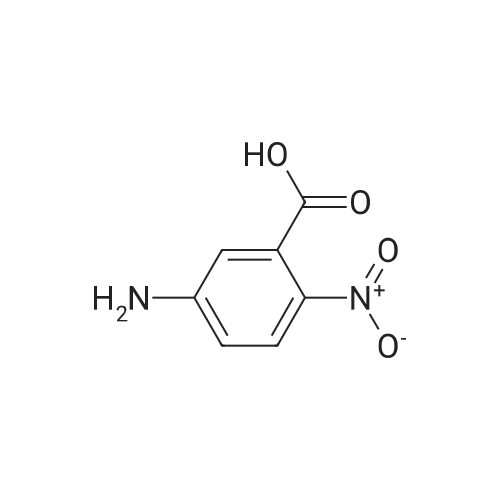
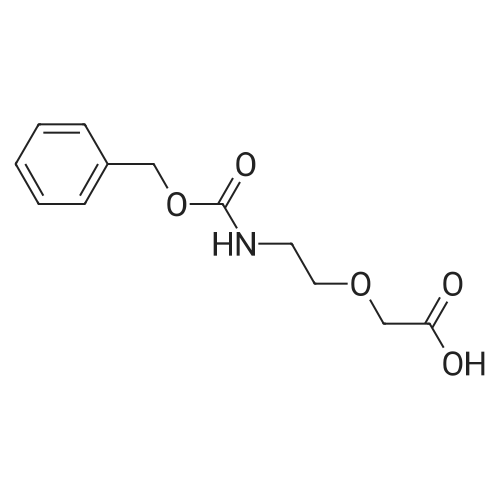
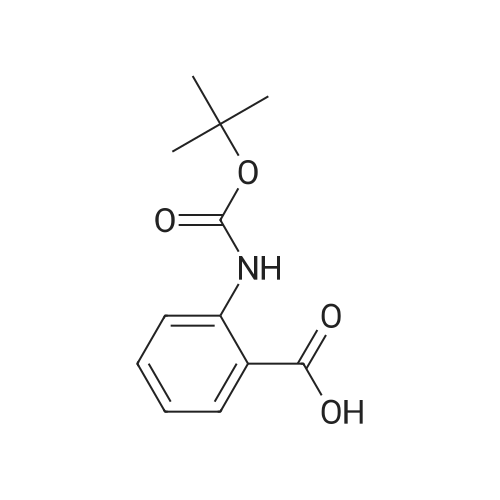
|
|
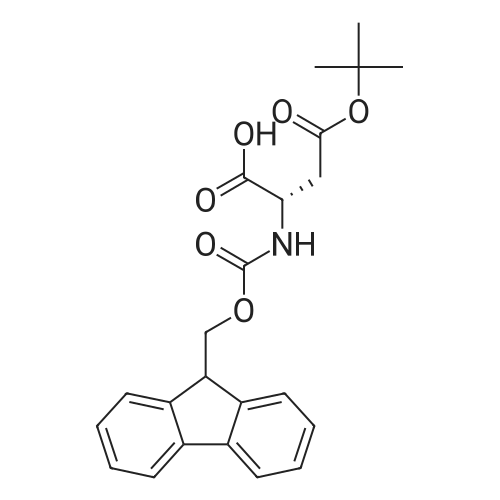
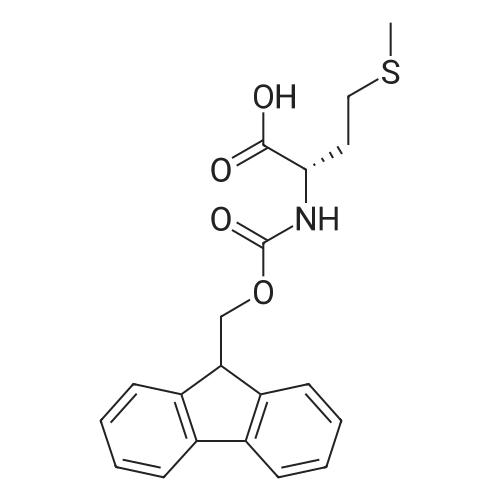
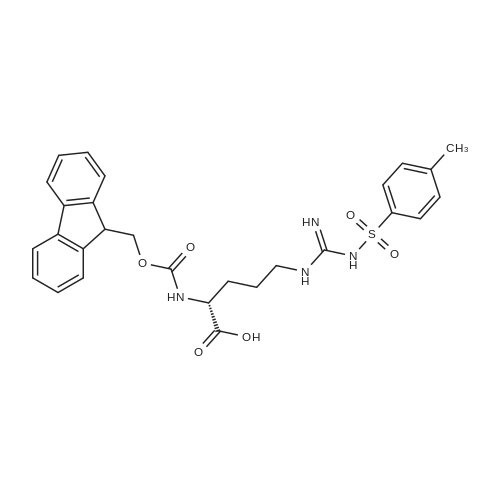
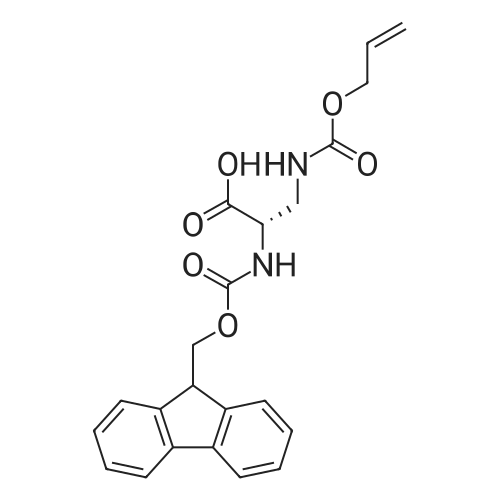
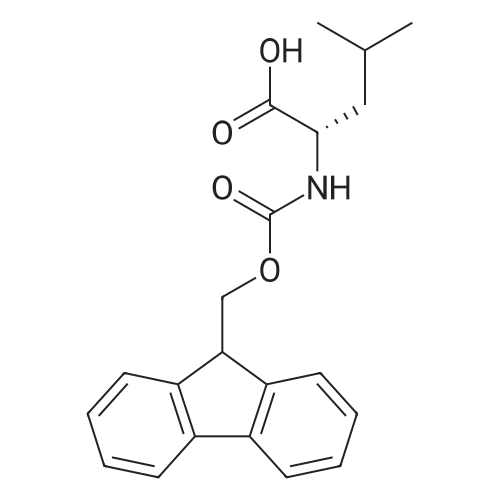
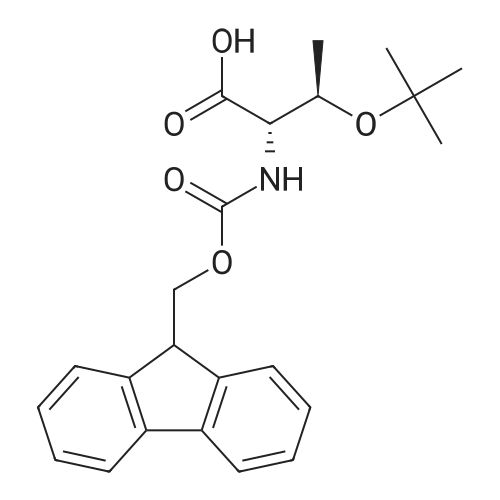
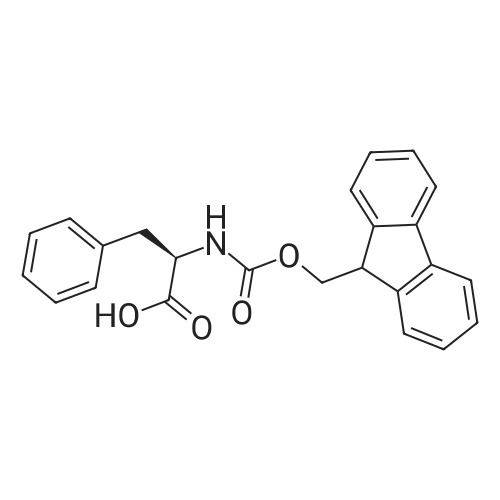
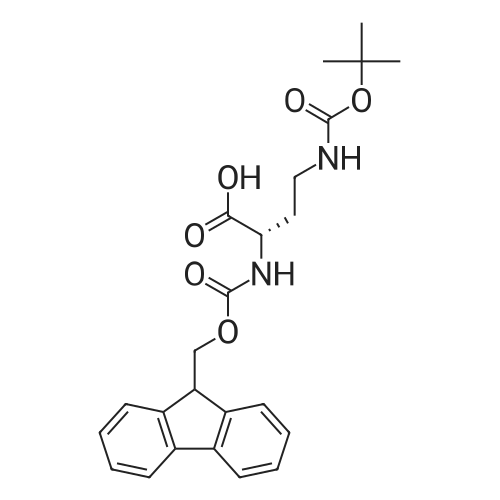
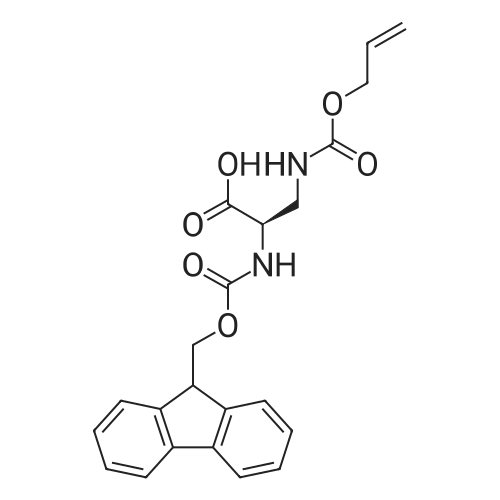
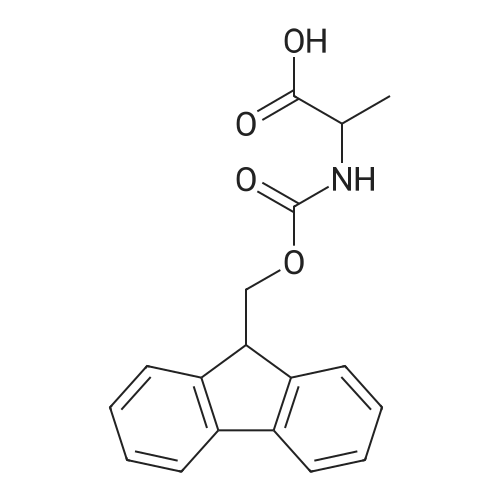
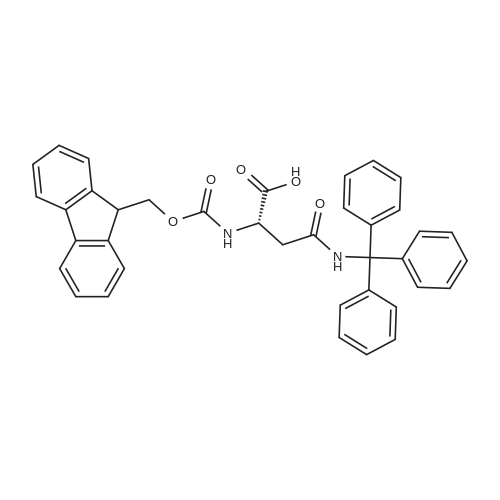
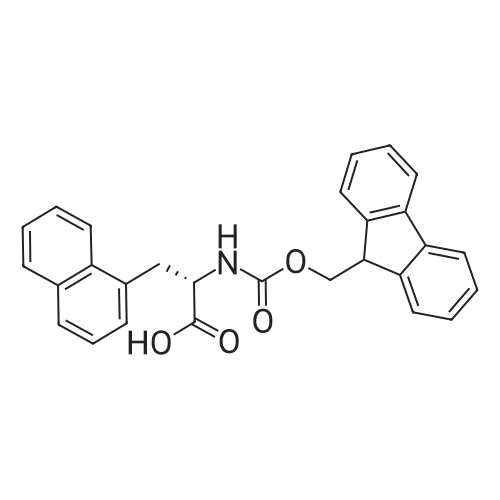
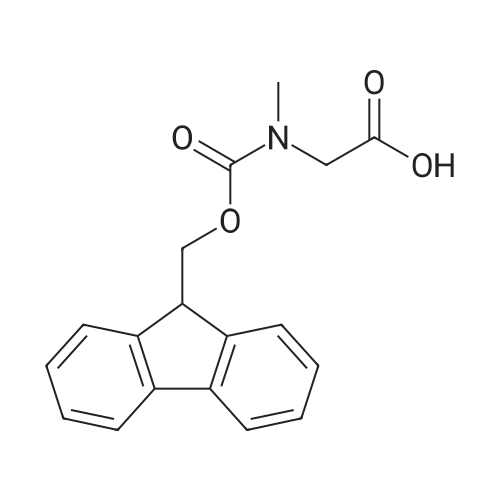
General procedure: Peptide synthesis was based on Fmoc chemistry, using a Symphony peptide synthesiser manufactured by Peptide Instruments and a Syro II synthesiser by MultiSynTech. Standard Fmoc- amino acids were employed (Sigma, Merck), with the following side chain protecting groups: Arg(Pbf); Asn(Trt); Asp(OtBu); Cys(Trt); GIu(OtBu); Gln(Trt); His(Trt); Lys(Boc); Ser(tBu); Thr(tBu); Trp(Boc); and Tyr(tBu) (Sigma). The coupling reagent was HCTU (Pepceuticals), diisopropylethylamine (DIPEA, Sigma) was employed as a base, and deprotection was achieved with 20percent piperidine in DMF (AGTC). Syntheses were performed using 0.37 mmol/gr Fmoc-Rink amide AM resin (AGTC), Fmoc-amino acids were utilised at a four-fold excess, and base was at a four-fold excess with respect to the amino acids. Amino acids were dissolved at 0.2M in DMSO, HCTU at 0.4M in DMF, and DIPEA at 1.6M in N-methylpyrrolidone (Alfa Aesar). Conditions were such that coupling reactions contained between 20 to 50percent DMSO in DMF, which reduced aggregation and deletions during the solid phase synthesis and enhanced yields. Coupling times were generally 30 minutes, and deprotection times 2 x 5 minutes. Fmoc-N-methylglycine (Fmoc- Sar-OH, Merck) was coupled for 1 hr, and deprotection and coupling times for the following residue were 20 min and 1 hr, respectively. After synthesis, the resin was washed with dichloromethane, and dried. Cleavage of side-chain protecting groups and from the support was effected using 10 mL of 95:2.5:2.5:2.5 v/v/v/w TFA/H20/iPr3SiH/dithiothreitol for 3 hours. Following cleavage, the spent resin was removed by filtration, and the filtrate was added to 35 mL of diethylether that had been cooled at -80°C. Peptide pellet was centrifuged, the etheric supernatant discarded, and the peptide pellet washed with cold ether two more times. Peptides were then resolubilised in 5-10 mL acetonitrile-water and lyophilised. A small sample was removed for analysis of purity of the crude product by mass spectrometry (MALDI-TOF, Voyager DE from Applied Biosystems). Following lyophilisation, peptide powders were taken up in 10 mL 6 M guanidinium hydrochloride in H20, supplemented with 0.5 mL of 1 M dithiothreitol, and loaded onto a C8 Luna preparative HPLC column (Phenomenex). Solvents (H20, acetonitrile) were acidified with 0.1 percent heptafluorobutyric acid. The gradient ranged from 30-70 percent acetonitrile in 15 minutes, at a flowrate of 15-20 mL /min, using a Gilson preparative HPLC system. Fractions containing pure linear peptide material (as identified by MALDI) were used for preparation of the bicycle derivatives by coupling to a scaffold molecule as described further below.A bicycle peptide designated 17-69-07-N434 was made corresponding to the bicycle peptide of Example lwith an N-terminal SarlO spacer similar to that of Reference Example 1, and conjugating group PYA (4-pentynoic acid, for "click" derivatisation with toxin). The structure of this derivative is shown schematically in Fig. 5. The linear peptide used to form this bicycle was as follows:(PYA)-(B-Ala)-SarlO-A(Dap)(D-Ala)NE(lNal)(D-Ala)CEDFYD(tBuGly)(Dap)The linear peptide and the bicycle peptide had the following LCMS Characteristics: |

 Chemistry
Chemistry
 Pharmaceutical Intermediates
Pharmaceutical Intermediates
 Inhibitors/Agonists
Inhibitors/Agonists
 Material Science
Material Science














 For Research Only
For Research Only
 120K+ Compounds
120K+ Compounds
 Competitive Price
Competitive Price
 1-2 Day Shipping
1-2 Day Shipping